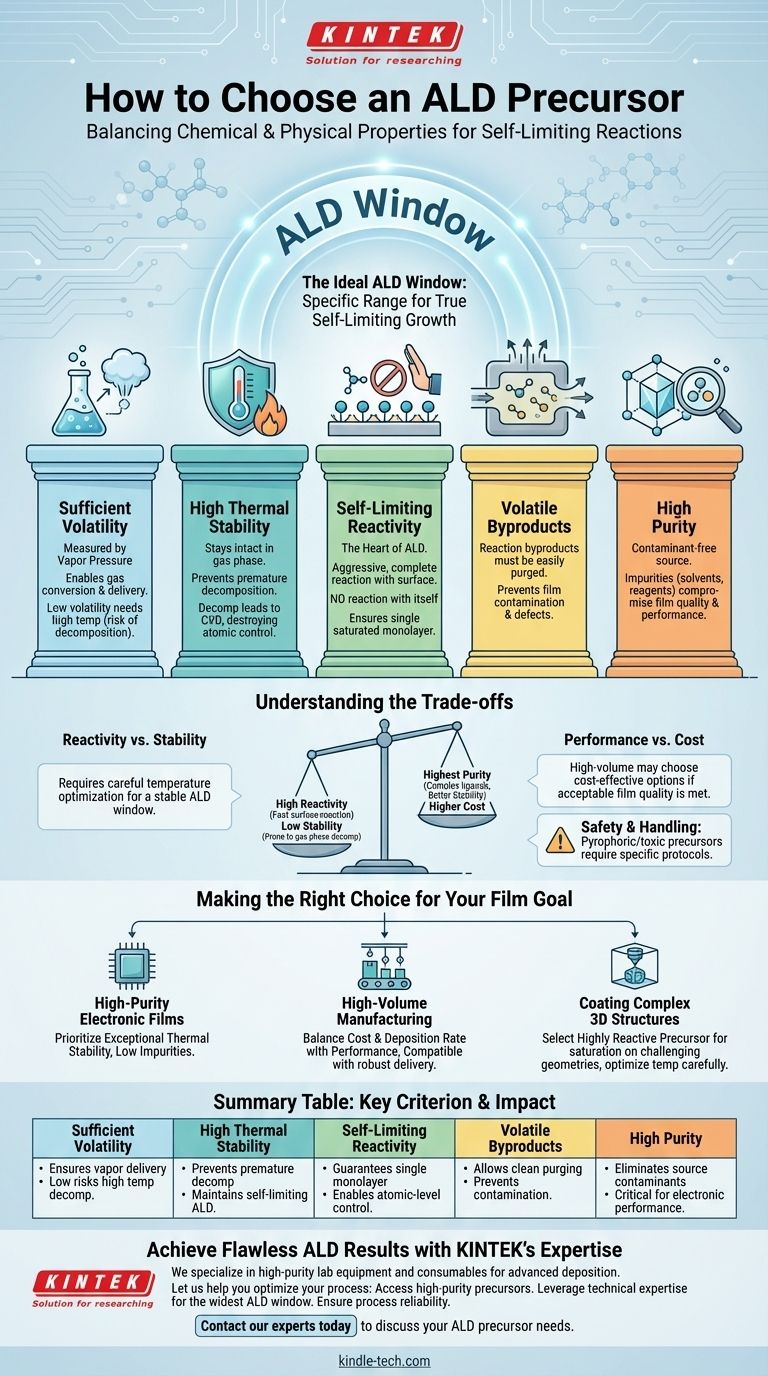
To choose the right ALD precursor, you must evaluate it based on a specific set of chemical and physical properties that enable the self-limiting reactions essential to Atomic Layer Deposition. The ideal precursor must have sufficient volatility to become a vapor, high thermal stability to prevent premature decomposition, and aggressive, self-limiting reactivity with the substrate. Furthermore, a precursor’s purity is non-negotiable, as contaminants directly compromise the quality and performance of the final thin film.
The core challenge in precursor selection is not simply finding a chemical that contains your desired element. It is about identifying a molecule whose combined properties of volatility, stability, and reactivity create a wide and reliable "ALD window"—the specific range of conditions where true self-limiting growth can occur.

The Foundation: Key Precursor Characteristics
The entire ALD process hinges on the predictable behavior of the precursor molecules. Each characteristic plays a critical role in whether a deposition will be successful, repeatable, and result in a high-quality film.
Sufficient Volatility
A precursor must be easily converted into a gas at a reasonable temperature and delivered into the reaction chamber. This is measured by its vapor pressure.
A precursor with low volatility requires high heating temperatures, which can complicate hardware design and potentially cause the molecule to decompose before it ever reaches the substrate.
High Thermal Stability
Once in the gas phase, the precursor must remain intact as it travels to the substrate. It should not break down due to heat alone.
If a precursor decomposes prematurely, the process degenerates from self-limiting ALD into continuous Chemical Vapor Deposition (CVD), destroying the atomic-level thickness control and film conformality.
Self-Limiting Reactivity
This is the heart of ALD. The precursor must react aggressively and completely with the active sites on the substrate surface, but it must not react with itself.
This ensures that only a single, saturated monolayer of the precursor is deposited in each cycle. The reaction must be complete to achieve uniform film growth.
Volatile Byproducts
The chemical reaction between the precursor and the surface generates byproduct molecules. These byproducts must also be volatile so they can be easily purged from the chamber.
If byproducts are not cleanly removed, they can be incorporated into the film as impurities, creating defects and degrading its electrical or optical properties.
High Purity
Any impurity in the precursor source—whether it's residual solvents, unreacted reagents, or molecules with different ligands—is a potential contaminant for your film.
For example, chloride impurities in a metal precursor can lead to chlorine incorporation in the final film, which can be corrosive and detrimental in semiconductor applications.
Understanding the Trade-offs
In practice, no precursor is perfect. The selection process often involves balancing competing properties and accepting certain compromises based on the specific application.
Reactivity vs. Stability
Often, the most highly reactive precursors are also the least thermally stable. A molecule that reacts very quickly with the surface may also be prone to decomposing in the gas phase if the process temperature fluctuates.
This trade-off forces a careful optimization of the deposition temperature to find a stable "ALD window."
Performance vs. Cost
The highest-purity precursors, especially those designed with complex organic ligands to enhance stability or volatility, can be significantly more expensive.
For high-volume manufacturing, a slightly less ideal but more cost-effective precursor might be chosen, provided the process can be optimized to still yield acceptable film quality.
Safety and Handling
Some of the most effective precursors can be pyrophoric (ignite spontaneously in air) or highly toxic. The choice of precursor is therefore constrained by the safety infrastructure and handling protocols available in the lab or factory.
Making the Right Choice for Your Film
Your final decision should be guided by the primary goal of your deposition process. Different priorities will lead you to weigh the precursor characteristics differently.
- If your primary focus is high-purity electronic films: Prioritize precursors with exceptional thermal stability and documented low levels of metallic, carbon, or halide impurities.
- If your primary focus is high-volume manufacturing: Balance precursor cost and deposition rate with performance, and consider precursors that are compatible with robust liquid delivery systems.
- If your primary focus is coating complex 3D structures: Select a highly reactive precursor that guarantees saturation even on challenging geometries, and be prepared to carefully optimize the temperature to avoid decomposition.
Ultimately, the best precursor is the one that provides the widest and most stable process window for your specific material and application.
Summary Table:
| Key Criterion | Why It Matters | Impact on ALD Process |
|---|---|---|
| Sufficient Volatility | Ensures the precursor can be delivered as a vapor. | Low volatility requires high temperatures, risking decomposition. |
| High Thermal Stability | Prevents premature decomposition in the gas phase. | Maintains self-limiting ALD growth vs. uncontrolled CVD. |
| Self-Limiting Reactivity | Guarantees a single, saturated monolayer per cycle. | Enables atomic-level thickness control and conformality. |
| Volatile Byproducts | Allows for clean purging from the reaction chamber. | Prevents film contamination and defects. |
| High Purity | Eliminates contaminants from the precursor source. | Critical for electronic film performance and reliability. |
Achieve Flawless ALD Results with KINTEK's Expertise
Selecting the ideal precursor is complex, but you don't have to do it alone. KINTEK specializes in providing high-purity lab equipment and consumables tailored for advanced deposition processes like ALD. We understand the critical balance between reactivity, stability, and cost for applications ranging from semiconductor fabrication to coating complex 3D structures.
Let us help you optimize your process:
- Access high-purity precursors that meet stringent electronic-grade standards.
- Leverage our technical expertise to identify the widest and most stable ALD window for your specific material.
- Ensure process reliability with consumables designed for consistent, high-performance results.
Ready to enhance your thin film quality and process yield? Contact our experts today to discuss your ALD precursor needs and find the perfect solution for your laboratory.
Visual Guide
Related Products
- Electrolytic Electrochemical Cell with Five-Port
- Laboratory Oscillating Orbital Shaker
- Thin-Layer Spectral Electrolysis Electrochemical Cell
- High Temperature Wear-Resistant Alumina Al2O3 Plate for Engineering Advanced Fine Ceramics
- Engineering Advanced Fine Ceramics Alumina Al2O3 Crucible With Lid Cylindrical Laboratory Crucible
People Also Ask
- What is galvanic cell or electrolytic cell? Unlock the Secrets of Electrochemical Power
- How does the design of an electrolytic cell affect the production yield of ferrate(VI)? Optimize Efficiency & Purity
- What are the key safety operation guidelines for using the electrolytic cell? Essential Protocols for Lab Safety
- What are the three essential components that comprise an electrolytic cell? Key Elements of Chemical Synthesis
- How can short circuits be prevented in the electrolytic cell setup? Essential Safety & Performance Tips