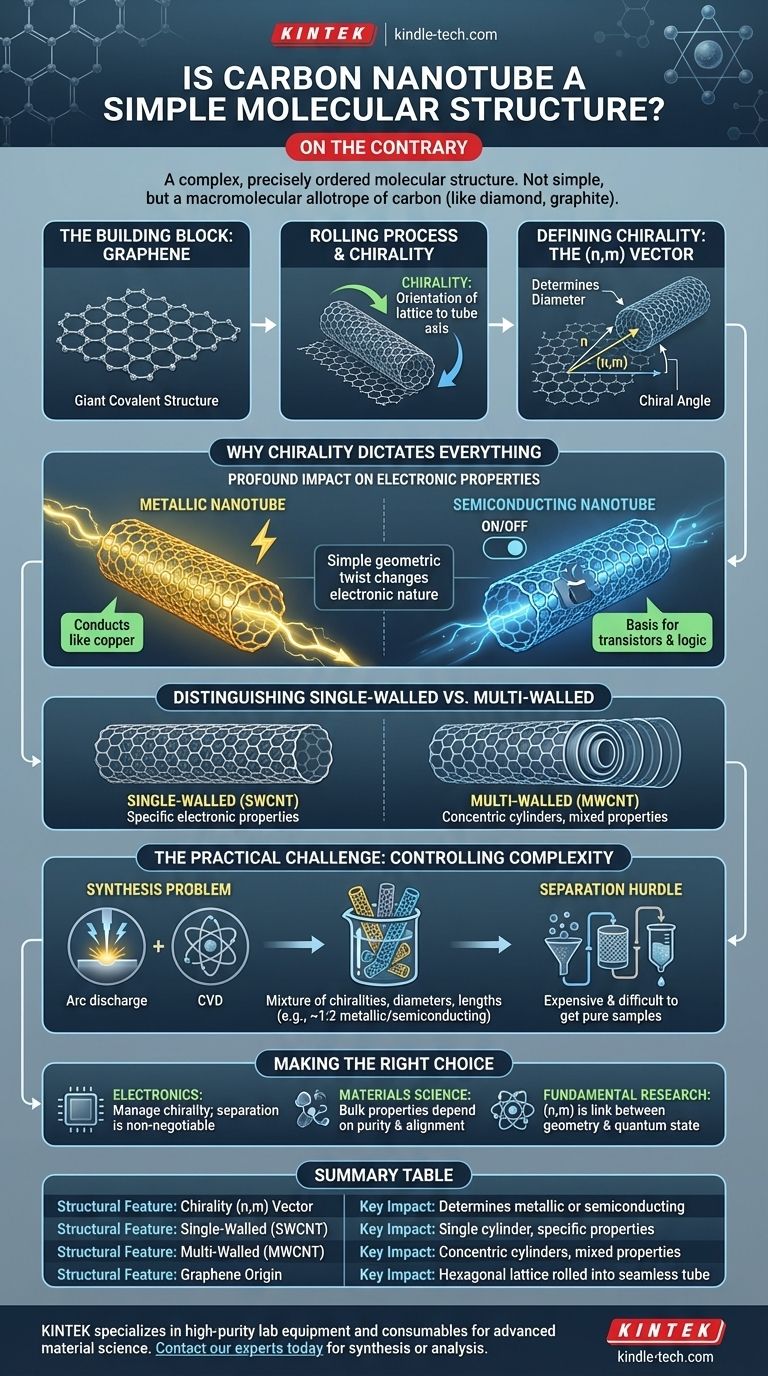
On the contrary, a carbon nanotube is an example of a highly complex and precisely ordered molecular structure. While the concept of a rolled-up sheet of carbon seems simple, its actual structure is defined by specific atomic arrangements that dictate its fundamental properties. It is not a simple molecule but a macromolecular allotrope of carbon, similar in class to diamond and graphite.
The core complexity of a carbon nanotube lies not in its size, but in its chirality—the exact angle at which the graphene sheet is "rolled" to form the tube. This single geometric parameter determines whether the nanotube will behave as a metal or a semiconductor, a distinction with profound implications.

From Graphene Sheet to Nanotube
To understand a nanotube's structure, you must first understand its origin. It is not assembled atom-by-atom in a random way but is derived from another complex carbon structure: graphene.
The Building Block: Graphene
A carbon nanotube is best visualized as a single sheet of graphene rolled into a seamless cylinder.
Graphene itself is a "giant covalent structure," a single layer of carbon atoms arranged in a honeycomb-like hexagonal lattice. Every atom is bonded to three others, creating a stable and strong plane.
The "Rolling" Process and Chirality
The term chirality is used to describe the orientation of the graphene lattice relative to the axis of the nanotube. Think of rolling a piece of paper with a hexagonal pattern on it. You can roll it straight, at an angle, or along a different axis.
Each of these "rolling" directions creates a nanotube with a different twist, or chirality. This is not a conceptual exercise; it represents a distinct and stable atomic arrangement.
Defining Chirality: The (n,m) Vector
This chirality is formally described by a pair of integers called the chiral vector, (n,m). This vector defines how the graphene sheet is rolled up.
Different (n,m) values correspond to different nanotube diameters and chiral angles, resulting in physically distinct structures. Two nanotubes with different (n,m) vectors are, for all practical purposes, different types of molecules.
Why Chirality Dictates Everything
The seemingly subtle difference in the (n,m) vector has massive consequences for the nanotube's physical and electronic properties. This is where the true complexity of the structure reveals itself.
The Profound Impact on Electronic Properties
This is the most critical consequence of chirality. Depending on the (n,m) indices, a carbon nanotube can have the electronic properties of a metal or a semiconductor.
A metallic nanotube conducts electricity like a copper wire. A semiconducting nanotube's ability to conduct electricity can be switched on and off, which is the fundamental requirement for building transistors and digital logic circuits.
The fact that a simple geometric twist in the atomic lattice completely changes its electronic nature is a unique and powerful feature of carbon nanotubes.
Distinguishing Single-Walled vs. Multi-Walled
To add another layer of structural complexity, nanotubes can exist as a single cylinder (Single-Walled Carbon Nanotube, or SWCNT) or as a series of concentric cylinders (Multi-Walled Carbon Nanotube, or MWCNT).
An MWCNT is like a set of Russian nesting dolls, where each nested tube can potentially have its own distinct chirality. This makes predicting the bulk electronic properties of MWCNTs even more complex.
The Practical Challenge: Controlling Complexity
The structural richness of carbon nanotubes is both a feature and a bug. While it offers a range of useful properties, it makes them incredibly difficult to work with.
The Synthesis Problem
The single greatest challenge in the field is controlled synthesis. Most production methods, like arc discharge or chemical vapor deposition, create a mixture of nanotubes with different chiralities, diameters, and lengths.
This means a typical batch of as-produced SWCNTs contains a mix of metallic and semiconducting types, often in a roughly 1:2 ratio.
The Separation Hurdle
For most high-value applications, particularly in electronics, a pure sample of either metallic or semiconducting nanotubes is required.
Separating this mixture is an expensive, difficult, and often inefficient process. This "complexity problem" is a major barrier to the widespread commercialization of nanotube-based electronics.
Making the Right Choice for Your Goal
Your perspective on a nanotube's structure depends entirely on your objective. Understanding its inherent complexity is the first step toward leveraging its properties effectively.
- If your primary focus is electronics: The key takeaway is that you must manage chirality. The presence of metallic tubes can short-circuit semiconducting-based devices, so separation or selective growth is non-negotiable.
- If your primary focus is materials science: The key takeaway is that the strong covalent bonds make all nanotubes exceptionally strong, but the bulk properties of a composite material will depend on the quality, purity, and alignment of the nanotube mixture.
- If your primary focus is fundamental research: The key takeaway is that the direct link between a simple geometric vector
(n,m)and the resulting quantum electronic state makes CNTs a perfect model system for studying nanoscale physics.
By appreciating that a carbon nanotube is not a single entity but a family of structures, you can navigate its challenges and exploit its extraordinary potential.
Summary Table:
| Structural Feature | Key Impact |
|---|---|
| Chirality (n,m) Vector | Determines if the nanotube is metallic or semiconducting |
| Single-Walled (SWCNT) | Single cylinder with specific electronic properties |
| Multi-Walled (MWCNT) | Concentric cylinders with potentially mixed properties |
| Graphene Origin | Hexagonal carbon lattice rolled into a seamless tube |
Ready to leverage carbon nanotubes in your research or production? KINTEK specializes in high-purity lab equipment and consumables for advanced material science. Whether you need controlled synthesis tools or analysis instruments for nanotube applications, our expertise ensures you get the precision and reliability your laboratory demands. Contact our experts today to discuss how we can support your innovative projects with the right equipment and consumables.
Visual Guide
Related Products
- Carbon Graphite Boat -Laboratory Tube Furnace with Cover
- Boron Nitride (BN) Ceramic Tube
- Hexagonal Boron Nitride HBN Thermocouple Protection Tube
- Hydrophilic Carbon Paper TGPH060 for Battery Lab Applications
People Also Ask
- What is the use of graphite tube? Essential for Extreme Heat & Corrosive Environments
- What metals can you melt in a graphite crucible? A Guide to Safe & Efficient Melting
- Does a graphite crucible need to be seasoned? The Critical First-Use Safety Guide
- Why is graphite used in furnaces? For Extreme Heat, Purity, and Efficiency
- What is the technical value of using graphite crucibles with graphite paper liners? Optimize Zr3(Al1-xSi)C2 Synthesis



