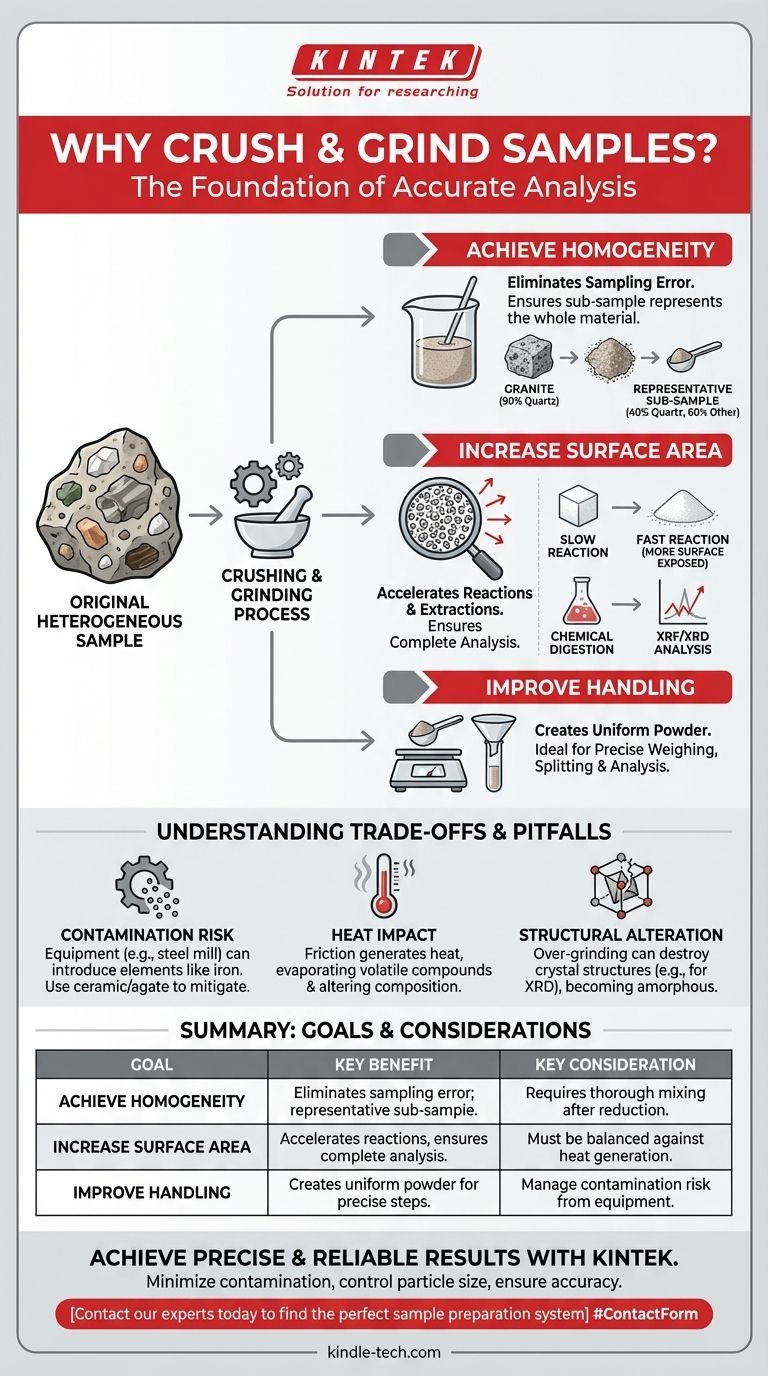
At its core, crushing and grinding samples is a foundational step to ensure the small portion you analyze is a true and accurate representation of the entire original material. This process achieves three primary goals: it homogenizes the sample, dramatically increases its surface area, and improves its handling characteristics for subsequent analysis or processing.
The fundamental purpose of sample reduction is not just to make large pieces small. It is to overcome the inherent heterogeneity of a material, guaranteeing that the final analytical result is reliable, repeatable, and truly reflects the composition of the whole.

The Principle of a Representative Sample
Most raw materials, from mineral ores to biological tissues, are not uniform. Analyzing a single piece would give a misleading result. Crushing and grinding is the mechanical process used to solve this problem.
The Challenge of Heterogeneity
Materials are often a mix of different components. A piece of granite, for example, contains distinct crystals of quartz, feldspar, and mica. Taking a single chip might yield a sample that is 90% quartz, while the rock as a whole is only 40% quartz. This is sampling error.
Achieving Homogeneity
By crushing a large sample into smaller particles and then mixing them thoroughly, you create a homogenized blend. In this state, any small sub-sample you take has a statistically identical composition to any other sub-sample. This ensures the portion you analyze is representative of the original bulk material.
The Power of Surface Area
Chemical reactions and physical analyses happen at the surface of a material. Grinding is the most effective way to maximize this active surface.
Accelerating Reactions and Extractions
Think of a sugar cube versus granulated sugar. The granulated sugar dissolves much faster because its total surface area exposed to the water is vastly greater. The same principle applies to laboratory work.
A fine powder allows solvents or acids to access and dissolve the target analyte much more quickly and completely during chemical digestion or leaching. This ensures you measure the total amount of a substance, not just what's on the surface of a larger chunk.
Ensuring Complete Analysis
Techniques like X-ray Fluorescence (XRF) or X-ray Diffraction (XRD) analyze a very small portion of the sample's surface. A coarse, unrepresentative sample will produce skewed data. A fine, homogeneous powder ensures the instrument's beam interacts with thousands of diverse particles, providing a true statistical average of the material's properties.
Understanding the Trade-offs and Pitfalls
While necessary, the process of crushing and grinding is not without risks that must be managed to ensure data integrity.
The Risk of Contamination
The grinding equipment itself can contaminate the sample. A steel mill can introduce iron, chromium, and nickel, which is a major problem for trace metal analysis. Using a ceramic or agate mortar and pestle can mitigate this, but even they can introduce trace amounts of silica or alumina.
The Impact of Heat
The friction of high-energy grinding generates significant heat. This can cause volatile compounds (like water or organic molecules) to evaporate, changing the sample's composition. Heat can also induce oxidation or other chemical changes in sensitive materials.
Altering Physical Structure
For certain analyses like XRD, the goal is to identify the material's crystal structure. Over-grinding can destroy this structure, creating an amorphous material and rendering the analysis useless. The process must be carefully controlled to reduce particle size without inducing structural damage.
Matching the Method to Your Objective
The right approach to sample preparation depends entirely on your end goal.
- If your primary focus is accurate chemical composition (e.g., elemental analysis, ore assaying): Your priority is creating a perfectly homogeneous powder to ensure the sub-sample is representative and has enough surface area for complete chemical digestion.
- If your primary focus is identifying crystalline phases (e.g., mineralogy via XRD): You must grind gently (e.g., by hand with a mortar and pestle) to create a fine powder without destroying the delicate crystal structures you intend to measure.
- If your primary focus is industrial processing (e.g., cement or pharmaceuticals): The goal is to achieve a specific particle size distribution that is optimal for the subsequent manufacturing step, such as ensuring a controlled reaction rate or product consistency.
Ultimately, precise and thoughtful sample preparation is the non-negotiable foundation for sound scientific and industrial results.
Summary Table:
| Goal of Crushing/Grinding | Key Benefit | Key Consideration |
|---|---|---|
| Achieve Homogeneity | Eliminates sampling error; ensures sub-sample represents the whole bulk material. | Requires thorough mixing after size reduction. |
| Increase Surface Area | Accelerates chemical reactions and extractions; ensures complete analysis. | Must be balanced against potential heat generation. |
| Improve Handling | Creates a uniform powder ideal for precise weighing, splitting, and instrumental analysis. | Risk of contamination from grinding equipment must be managed. |
Achieve precise and reliable results in your lab. Proper sample preparation is the critical first step. KINTEK specializes in high-quality lab equipment and consumables for crushing, grinding, and homogenizing your samples. Our solutions help you minimize contamination, control particle size, and ensure your analysis is accurate every time.
Contact our experts today to find the perfect sample preparation system for your specific application, from chemical assay to XRD analysis.
Visual Guide
Related Products
- Laboratory Grinding Mill Mortar Grinder for Sample Preparation
- Laboratory Disc Cup Vibratory Mill for Sample Grinding
- Laboratory Sealed Hammer Crusher for Efficient Sample Preparation
- Laboratory Hybrid Tissue Grinding Mill
- Powerful Plastic Crusher Machine
People Also Ask
- What does a grinder do in a laboratory? Achieve Homogeneous Sample Preparation for Accurate Analysis
- What laboratory apparatus is used for grinding? Match the Right Mill to Your Sample Material
- What are the different types of laboratory mills? Choose the Right Grinder for Your Sample Material
- What is the purpose of using a laboratory grinder and standard sieves? Unlock Precise Rice Husk Pretreatment
- What is the function of a laboratory grinding mill? Achieve Precise Sample Preparation for Accurate Results