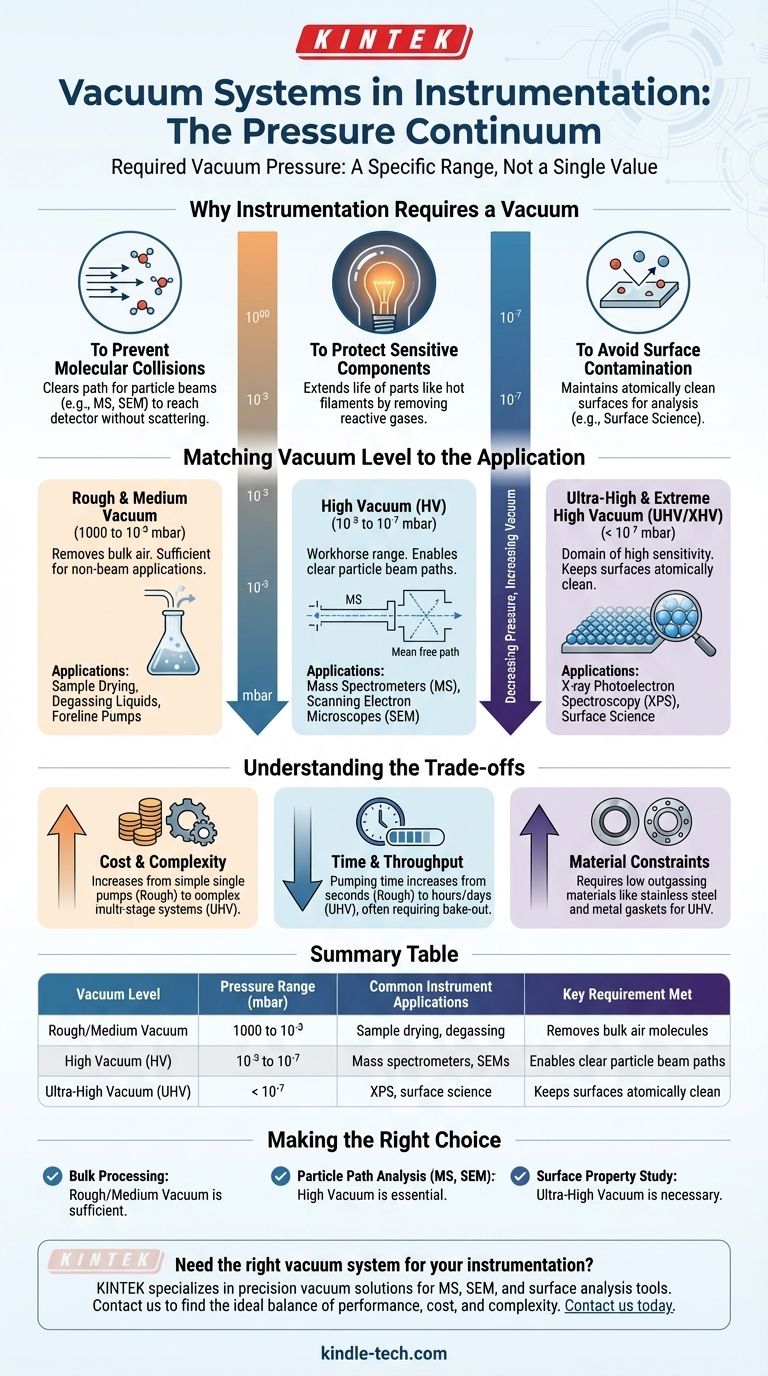
The required vacuum pressure for an instrument is not a single value but rather a specific range dictated entirely by the instrument's function. This can span from a Rough Vacuum (around 1 mbar) for sample preparation to an Ultra-High Vacuum (below 10⁻⁷ mbar) for sensitive surface analysis, with many analytical instruments operating in the High Vacuum range (10⁻³ to 10⁻⁷ mbar).
The core principle is straightforward: the level of vacuum required is determined by the need to eliminate interference from air molecules. The "better" the vacuum (lower the pressure), the fewer molecules remain, and the less likely they are to collide with the particles or samples you are trying to measure.

Why Instrumentation Requires a Vacuum
At its heart, a vacuum system is designed to create a controlled environment by removing atmospheric gas molecules. Different instruments require this control for different reasons, all of which are critical for generating accurate data.
To Prevent Molecular Collisions
Many instruments, like mass spectrometers or electron microscopes, work by accelerating a beam of particles (ions or electrons) from a source to a detector.
In normal air pressure, this beam would immediately collide with billions of nitrogen, oxygen, and other gas molecules. These collisions would scatter the beam, cause unwanted chemical reactions, and make any measurement impossible. A vacuum ensures the particles have a clear, unobstructed path.
To Protect Sensitive Components
Certain components, like the hot filaments used to generate electrons in an electron microscope, would instantly burn out (oxidize) if exposed to oxygen at high temperatures.
A vacuum environment removes the reactive gases, dramatically extending the life and stability of these critical parts.
To Avoid Surface Contamination
For instruments that analyze material surfaces (like surface science techniques), any residual gas molecules in the chamber will quickly stick to and contaminate the sample.
An ultra-high vacuum is necessary to keep a surface atomically clean long enough for an analysis to be completed.
Matching Vacuum Level to the Application
The specific pressure range an instrument needs is directly tied to how much molecular interference it can tolerate. This is why vacuum levels are categorized into distinct regimes.
Rough & Medium Vacuum (1000 to 10⁻³ mbar)
This level of vacuum removes the vast majority of air molecules but still leaves a significant number behind.
It is sufficient for applications like sample drying, degassing liquids, or serving as the initial "foreline" pressure for more powerful high-vacuum pumps. It is not suitable for instruments with particle beams.
High Vacuum (HV) (10⁻³ to 10⁻⁷ mbar)
This is the workhorse range for a vast number of analytical instruments, including most mass spectrometers (MS) and scanning electron microscopes (SEM).
At these pressures, the average distance a molecule can travel before hitting another (the mean free path) becomes longer than the dimensions of the instrument's chamber. This ensures that particles can travel from the source to the detector without collision, enabling accurate measurement.
Ultra-High & Extreme High Vacuum (UHV/XHV) (< 10⁻⁷ mbar)
This is the domain of highly sensitive surface science and semiconductor manufacturing.
At these extremely low pressures, it can take minutes, hours, or even days for a single layer of gas molecules to form on a pristine surface. This gives researchers the time needed to perform detailed analyses on uncontaminated samples using techniques like X-ray Photoelectron Spectroscopy (XPS).
Understanding the Trade-offs
Choosing a vacuum level is not simply about aiming for the lowest possible pressure. Higher levels of vacuum introduce significant practical challenges.
Cost and Complexity
Achieving a rough vacuum requires a single, relatively inexpensive mechanical pump. Achieving UHV requires a multi-stage system with several pumps (e.g., mechanical, turbomolecular, and ion pumps), specialized all-metal components, and complex control systems, making it orders of magnitude more expensive.
Time and Throughput
A rough vacuum can be achieved in seconds or minutes. Pumping a system down to high vacuum might take an hour. Reaching UHV can take many hours or even days, often requiring the entire system to be "baked out" at high temperatures to drive off adsorbed water and gas molecules from the chamber walls.
Material Constraints
Low-vacuum systems can use simple rubber O-rings and flexible materials. UHV systems demand stainless steel construction, metal gaskets (like copper), and materials with very low outgassing rates to avoid being a source of contamination themselves.
Making the Right Choice for Your Goal
The correct vacuum pressure is the one that meets the minimum requirements for your measurement without adding unnecessary cost and complexity.
- If your primary focus is bulk material processing like drying or degassing: A rough or medium vacuum is sufficient and highly cost-effective.
- If your primary focus is analyzing particle paths, like in a standard mass spectrometer or SEM: High vacuum is the non-negotiable standard to ensure a clear path from source to detector.
- If your primary focus is studying the fundamental properties of an atomically clean surface: Ultra-high vacuum is essential to provide a pristine environment free from atmospheric contamination.
Ultimately, selecting the right vacuum level is about creating an environment where your instrument can perform its measurement reliably and without interference.
Summary Table:
| Vacuum Level | Pressure Range (mbar) | Common Instrument Applications | Key Requirement Met |
|---|---|---|---|
| Rough/Medium Vacuum | 1000 to 10⁻³ | Sample drying, degassing | Removes bulk air molecules |
| High Vacuum (HV) | 10⁻³ to 10⁻⁷ | Mass spectrometers, SEMs | Enables clear particle beam paths |
| Ultra-High Vacuum (UHV) | < 10⁻⁷ | XPS, surface science | Keeps surfaces atomically clean |
Need the right vacuum system for your instrumentation? KINTEK specializes in lab equipment and consumables, serving laboratory needs with precision vacuum solutions for mass spectrometers, SEMs, and surface analysis tools. Our experts will help you select the ideal system that balances performance, cost, and complexity for your specific application. Contact us today to ensure your measurements are accurate and interference-free!
Visual Guide
Related Products
- Vacuum Induction Melting Spinning System Arc Melting Furnace
- CF KF Flange Vacuum Electrode Feedthrough Lead Sealing Assembly for Vacuum Systems
- Circulating Water Vacuum Pump for Laboratory and Industrial Use
- Vacuum Bellows for Efficient Connection and Stable Vacuum in High-Performance Systems
- 304 316 Stainless Steel Vacuum Ball Valve Stop Valve for High Vacuum Systems
People Also Ask
- What is the principle of vacuum induction melting? Achieve Ultra-High Purity Metals
- What are the key components inside the vacuum chamber of a vacuum induction melting furnace? A Guide to the Core Melting Assembly
- How does vacuum induction melting work? Achieve Ultra-Pure, High-Performance Alloys
- What are the advantages of a vacuum induction melting furnace? Achieve High-Purity Alloys with Precision VIM
- How do vacuum induction or arc melting furnaces facilitate the synthesis of U-Al-C MAX phases? Precision Heat & Purity



















