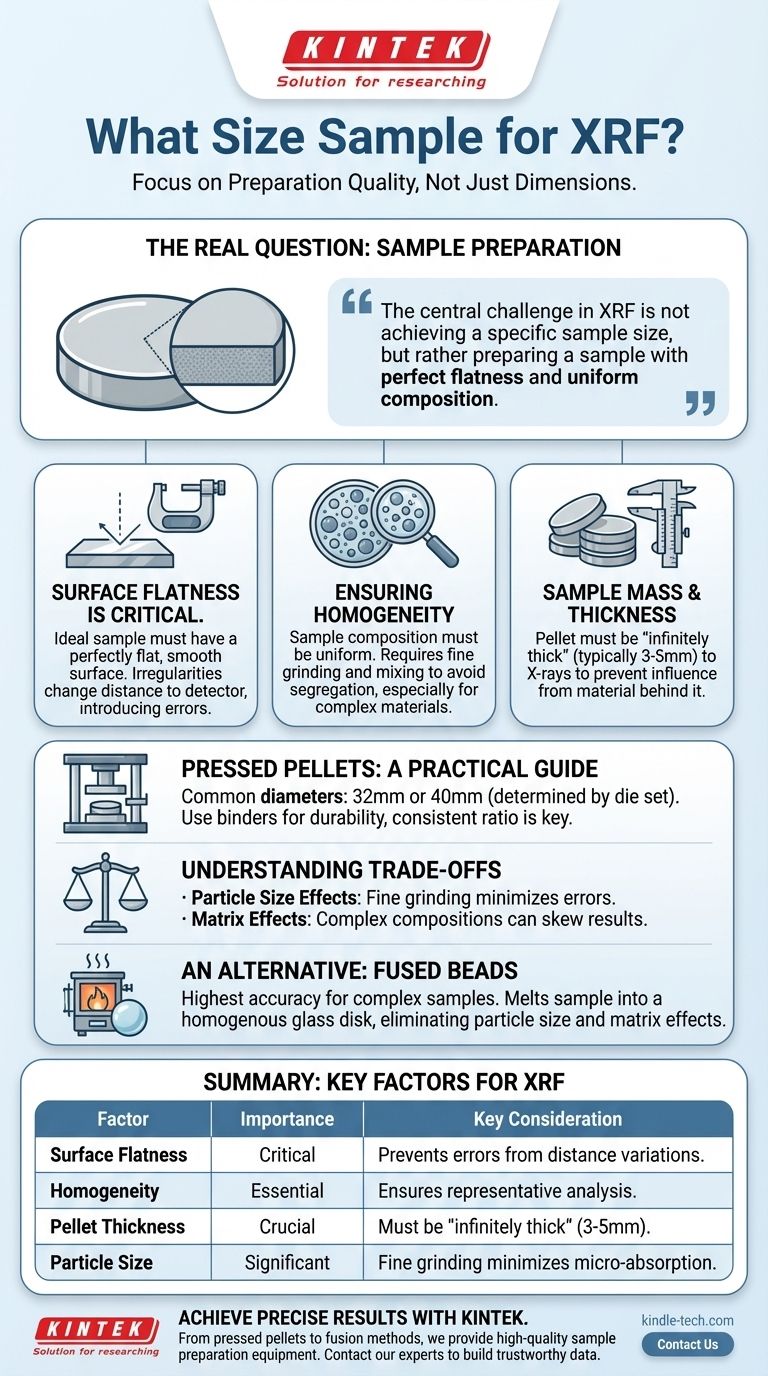
For X-Ray Fluorescence (XRF) analysis, the most common sample form is a pressed pellet, typically 32mm or 40mm in diameter. However, the physical diameter is far less important than other factors like surface flatness, sample homogeneity, and sufficient thickness to ensure the analysis is accurate and repeatable.
The central challenge in XRF is not achieving a specific sample size, but rather preparing a sample with perfect flatness and uniform composition. The quality of your preparation directly dictates the quality of your results.

Why Sample Preparation is the Real Question
Your question about "size" points to a deeper need: ensuring your sample is prepared correctly for analysis. An improperly prepared sample, regardless of its dimensions, will produce unreliable data. The goal is to present a perfectly uniform and representative face to the X-ray beam.
The Critical Role of a Flat Surface
An ideal sample for XRF must have a perfectly flat, smooth surface. XRF instruments are calibrated for a precise distance between the X-ray source, the sample, and the detector.
Any surface irregularities, bumps, or voids change this critical distance. This variation alters the intensity of the fluorescent X-rays reaching the detector, introducing significant errors into your elemental analysis.
Ensuring Homogeneity
The analyzed area is small, so the sample must be homogenous, meaning its composition is uniform throughout. If the material has large or uneven particles, you risk analyzing an area that isn't representative of the whole.
For many materials, this requires grinding them into a fine powder. For more complex samples, initial processing with equipment like a jaw crusher may be necessary to achieve a uniform particle size before pressing.
The Pressed Pellet Method: A Practical Guide
Creating pressed pellets is a common, cost-effective, and reliable method for preparing solid samples for XRF. The "size" is determined by the equipment you use, but the quality is determined by your technique.
Pellet Diameter (The "Size")
The diameter of the pellet is determined by the die set used in your hydraulic press. Standard sizes are typically 32mm or 40mm, which fit into the sample holders of most commercial XRF spectrometers.
Sample Mass and Thickness
The amount of powdered sample you use determines the final thickness of the pellet. The pellet must be "infinitely thick" to the X-ray beam, meaning it's thick enough that the primary X-rays cannot pass all the way through it.
If a sample is too thin, the analysis will be influenced by the material behind or beneath the sample, leading to incorrect readings. A good starting point for many materials is a final pellet thickness of 3-5mm.
Binders and Dilution
Often, a binder or grinding aid is mixed with the sample powder. This helps the particles stick together during pressing, resulting in a durable, crack-free pellet.
The choice of binder and the sample-to-binder ratio are critical parts of your preparation recipe. This ratio must be consistent across all samples and standards to ensure comparability.
Understanding the Trade-offs
While pressed pellets are a standard method, they are not without limitations. Understanding these helps you avoid common pitfalls and decide if a more advanced technique is required.
Particle Size Effects
Even with fine grinding, micro-absorption effects related to particle size can skew results, particularly for lighter elements. Inhomogeneous particle sizes can lead to segregation, where finer or denser particles settle unevenly in the die before pressing.
Matrix Effects
The overall composition of the sample (the "matrix") can affect the intensity of the fluorescent X-rays from the elements you want to measure. For highly variable or complex materials, pressed pellets may not be sufficient to overcome these matrix effects, leading to inaccuracies.
An Alternative: Fused Beads
For the highest accuracy, especially with complex geological or industrial samples, fusion is the preferred method. This involves melting the sample with a lithium borate flux at high temperatures to create a perfectly homogenous glass disk. This process eliminates particle size and mineralogical effects but requires specialized equipment like fusion furnaces and platinum labware.
Making the Right Choice for Your Goal
Your analytical needs should dictate your preparation method. Focus on creating a consistent, repeatable process to generate data you can trust.
- If your primary focus is routine, high-throughput analysis of similar materials: The pressed pellet method offers an excellent balance of speed, cost, and quality.
- If your primary focus is high-accuracy analysis of complex or unknown materials: You should strongly consider the fusion method to eliminate matrix and particle size effects.
Ultimately, proper sample preparation is the foundation of trustworthy XRF analysis.
Summary Table:
| Factor | Importance for XRF | Key Consideration |
|---|---|---|
| Surface Flatness | Critical | Prevents measurement errors from distance variations. |
| Homogeneity | Essential | Ensures the analyzed spot is representative of the whole sample. |
| Pellet Thickness | Crucial | Must be "infinitely thick" (typically 3-5mm) to avoid interference. |
| Particle Size | Significant | Fine grinding minimizes micro-absorption effects. |
Achieve precise and reliable XRF results with expert sample preparation solutions from KINTEK.
Whether your lab focuses on high-throughput analysis with pressed pellets or requires the ultimate accuracy of the fusion method, the right equipment is crucial. KINTEK specializes in high-quality lab equipment, including hydraulic presses, dies, and fusion furnaces, to meet the specific needs of your laboratory.
Don't let sample preparation be the weak link in your analytical workflow. Contact our experts today to discuss how we can help you build a foundation of trustworthy data.
Visual Guide
Related Products
- XRF Boric Acid Lab Powder Pellet Pressing Mold for Laboratory Use
- XRF & KBR plastic ring lab Powder Pellet Pressing Mold for FTIR
- XRF & KBR steel ring lab Powder Pellet Pressing Mold for FTIR
- Laboratory Hydraulic Pellet Press for XRF KBR FTIR Lab Applications
- Automatic Laboratory Hydraulic Press for XRF & KBR Pellet Press
People Also Ask
- What is the XRF pressed pellet method? A Fast, Cost-Effective Sample Prep Guide
- How do you prepare a pellet press sample for XRF? A Step-by-Step Guide to Accurate Analysis
- How are samples prepared for XRF analysis? Achieve Accurate and Reliable Results
- How do you make XRF pellets? A 4-Step Guide to Flawless Sample Preparation
- How does a laboratory powder pellet press facilitate the preparation of multi-layered gradient Al2O3/ZrO2 ceramic green bodies? Precision Bonding Techniques
















