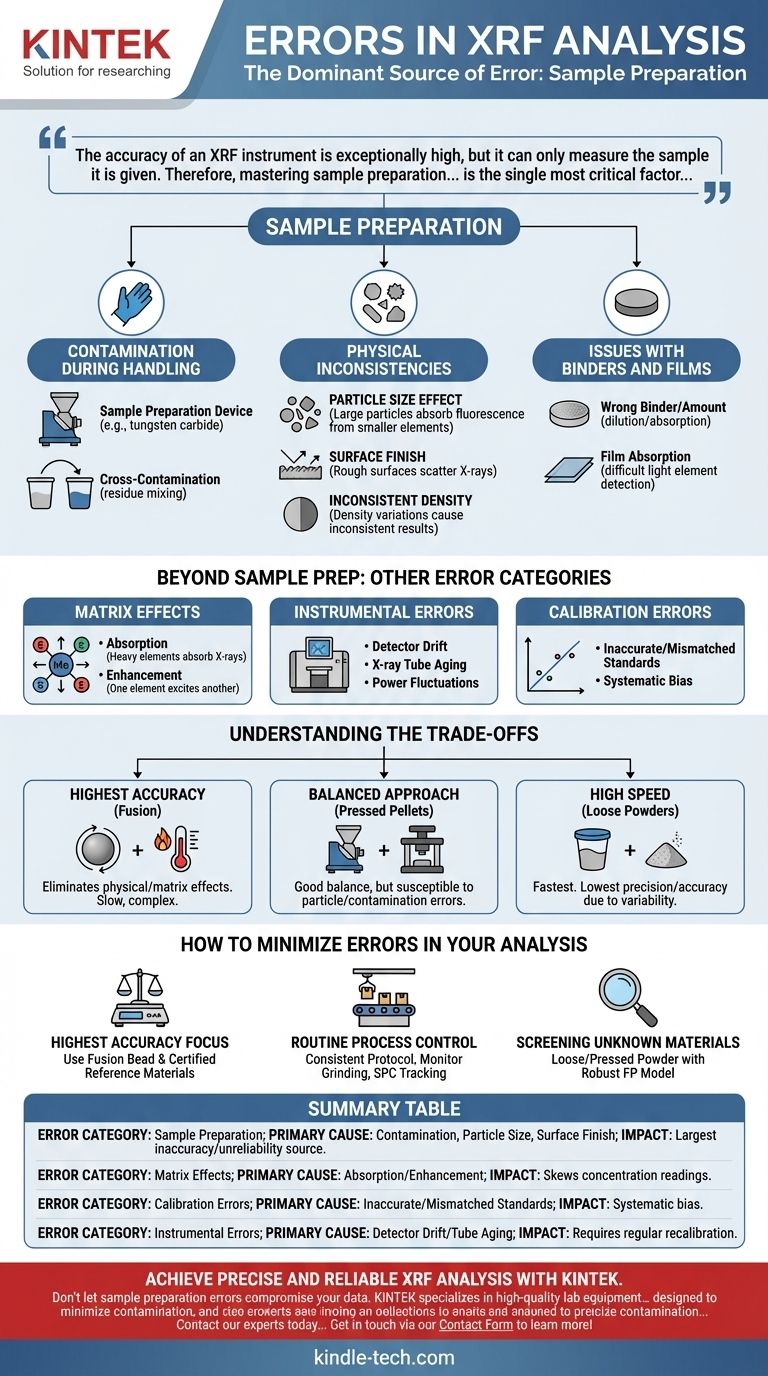
In modern X-ray Fluorescence (XRF) analysis, the most significant source of error is no longer the instrument itself but the sample being measured. While instrumental and matrix effects exist, inconsistencies and contamination introduced during sample preparation are the primary cause of inaccurate and unreliable results.
The accuracy of an XRF instrument is exceptionally high, but it can only measure the sample it is given. Therefore, mastering sample preparation and understanding its impact is the single most critical factor in achieving trustworthy analytical data.

The Dominant Source of Error: Sample Preparation
The capabilities of modern XRF spectrometers, in terms of stability and sensitivity, have advanced to a point where the sample has become the weakest link in the analytical chain.
Why Sample Preparation is Critical
XRF is a comparative and surface-sensitive technique. The instrument assumes the small area it analyzes is a perfect representation of the entire sample. Any deviation from this ideal state, whether chemical or physical, introduces error.
Proper preparation aims to present a sample to the spectrometer that is homogeneous, flat, and representative of the bulk material.
Contamination During Handling
Contamination can occur at multiple stages, but the grinding process is the most common source.
It typically arises from two places: the sample preparation device (e.g., tungsten carbide from a grinding mill) or sample-to-sample cross-contamination, where residue from a previous sample is mixed into the current one.
Physical Inconsistencies
For powdered samples, physical properties create significant errors.
- Particle Size Effect: X-rays penetrate the sample to a finite depth. If particles are too large, the X-ray fluorescence from smaller or lighter elements can be disproportionately absorbed by larger, heavier particles, skewing the results.
- Surface Finish: A rough or uneven surface on a pressed pellet can scatter X-rays unpredictably, reducing the signal intensity that reaches the detector.
- Inconsistent Density: A poorly pressed pellet with density variations will lead to inconsistent analytical results across its surface.
Issues with Binders and Films
Binders are used to hold pressed pellets together, and thin films are used to contain loose powders or liquids. While necessary, they can be a source of error.
Using the wrong binder or an incorrect amount can dilute the sample or absorb the fluorescence of the target elements, leading to systematically low readings. The film itself can also absorb low-energy X-rays from light elements, making them difficult to detect.
Beyond Sample Prep: Other Error Categories
While sample preparation is the largest variable, a comprehensive understanding requires acknowledging other error sources that are inherent to the technique or the instrument.
Matrix Effects
The "matrix" refers to everything in the sample that is not the element being measured. These other elements can interfere with the analysis.
- Absorption: A heavy element in the matrix can absorb the characteristic X-rays emitted by a lighter target element, making the concentration of the light element appear lower than it is.
- Enhancement: An element in the matrix can emit X-rays that, in turn, excite a target element, causing it to fluoresce more strongly. This makes the concentration of the target element appear higher than it is.
Instrumental Errors
Although minor in modern, well-maintained instruments, errors can still arise from the hardware itself. These include detector drift over time, X-ray tube aging (which reduces output), and minor power supply fluctuations. Regular performance testing and recalibration are used to correct for these.
Calibration Errors
XRF relies on a calibration model to convert raw X-ray counts into elemental concentrations. If the standards used to build this calibration are inaccurate or not sufficiently similar to the unknown samples, the entire analysis will be systematically flawed. This is a critical source of bias.
Understanding the Trade-offs
Choosing a sample preparation method involves a fundamental trade-off between accuracy, speed, and cost.
- Highest Accuracy (Fusion): Creating a fused bead by dissolving the sample in a flux at high temperature eliminates all particle size and mineralogical effects. This is the gold standard for accuracy but is also slow, complex, and requires expensive equipment.
- Balanced Approach (Pressed Pellets): Grinding a sample and pressing it into a pellet is the most common method. It offers a good balance of accuracy and throughput but is susceptible to the particle size and contamination errors discussed earlier.
- High Speed (Loose Powders): Analyzing a loose powder in a sample cup is very fast. However, it provides the lowest precision and accuracy due to variable density and surface effects. It is best reserved for qualitative screening or when only semi-quantitative data is needed.
How to Minimize Errors in Your Analysis
Your strategy for minimizing error depends entirely on your analytical goal. Acknowledge the largest potential error source—the sample—and allocate your effort accordingly.
- If your primary focus is the highest possible accuracy: Use the fusion bead method to eliminate physical and matrix effects, and calibrate with certified reference materials.
- If your primary focus is routine process control: Develop a highly consistent pressed powder protocol, monitor grinder contamination, and use statistical process control to track instrument drift.
- If your primary focus is screening unknown materials: Use a loose powder or simple pressed pellet method with a robust Fundamental Parameters (FP) software model, but always be aware of its inherent limitations.
Ultimately, achieving reliable XRF results comes from understanding and controlling the variables before the measurement even begins.
Summary Table:
| Error Category | Primary Cause | Impact on Results |
|---|---|---|
| Sample Preparation | Contamination, inconsistent particle size, poor surface finish | Largest source of inaccuracy and unreliability |
| Matrix Effects | Absorption or enhancement of X-rays by other elements | Skews concentration readings (high or low) |
| Calibration Errors | Inaccurate or mismatched standards | Systematic bias across all analyses |
| Instrumental Errors | Detector drift, tube aging (minor in modern instruments) | Requires regular performance testing and recalibration |
Achieve precise and reliable XRF analysis with KINTEK.
Don't let sample preparation errors compromise your data. KINTEK specializes in high-quality lab equipment and consumables—including mills, presses, pellets, and fusion flux—designed to minimize contamination and ensure consistent, representative samples.
Contact our experts today to discuss your specific laboratory needs and discover how our solutions can enhance your analytical accuracy.
Get in touch via our Contact Form to learn more!
Visual Guide
Related Products
- XRF Boric Acid Lab Powder Pellet Pressing Mold for Laboratory Use
- XRF & KBR plastic ring lab Powder Pellet Pressing Mold for FTIR
- XRF & KBR steel ring lab Powder Pellet Pressing Mold for FTIR
- Laboratory Hydraulic Pellet Press for XRF KBR FTIR Lab Applications
- Automatic Laboratory Hydraulic Press for XRF & KBR Pellet Press
People Also Ask
- How do you make XRF pellets? A 4-Step Guide to Flawless Sample Preparation
- What is the purpose of using a mold for pellet pressing when preparing catalyst test samples? Ensure Data Consistency
- How do you prepare a pellet press sample for XRF? A Step-by-Step Guide to Accurate Analysis
- How do you prepare a sample for XRF? Achieve Accurate and Reliable Analysis
- How does the selection of a pressure mold affect the performance of all-solid-state batteries? Expert Pelletizing Guide










