Requirements for the Use of Reference Electrodes
Ideal Non-Polarized Electrode
An ideal non-polarized electrode serves as a cornerstone in electrochemical measurements, providing a stable and reliable reference point. This type of electrode is characterized by its excellent reversibility, which ensures that the electrode potential remains consistent regardless of the direction of the current flow. A stable potential is crucial, as it minimizes fluctuations and errors in measurements, thereby enhancing the accuracy and reliability of the data obtained.
Additionally, the potential reproducibility of an ideal non-polarized electrode is paramount. This means that the electrode should consistently produce the same potential under identical conditions, eliminating discrepancies in repeated experiments. Such reproducibility is essential for scientific rigor and for ensuring that experimental results can be reliably compared and replicated.
To illustrate these qualities, consider the following table comparing the characteristics of ideal and non-ideal reference electrodes:
| Characteristic | Ideal Reference Electrode | Non-Ideal Reference Electrode |
|---|---|---|
| Electrode Reversibility | High | Low |
| Potential Stability | High | Variable |
| Potential Reproducibility | High | Low |
In summary, an ideal non-polarized electrode must exhibit high electrode reversibility, stable potential, and excellent potential reproducibility to function effectively as a reference electrode in electrochemical studies.
Temperature Coefficient and Maintenance
The temperature coefficient of a reference electrode is a critical parameter that influences its performance and reliability. Ideally, the electrode should exhibit a minimal temperature coefficient, ensuring that its potential remains stable across varying environmental conditions. This stability is crucial for accurate measurements in electrochemical experiments, where even minor fluctuations in potential can lead to significant errors.
In addition to a small temperature coefficient, the electrode must be user-friendly in terms of fabrication, operation, and maintenance. The ease of manufacturing ensures consistent quality and availability, while straightforward usage simplifies the experimental setup and reduces the likelihood of operational errors. Maintenance requirements should be minimal, allowing for long-term use without frequent calibration or replacement.
| Aspect | Ideal Characteristics | Importance |
|---|---|---|
| Temperature Coefficient | Small | Ensures stable potential across varying temperatures |
| Fabrication | Easy to produce | Ensures consistent quality and availability |
| Usage | Simple and intuitive | Simplifies experimental setup and reduces errors |
| Maintenance | Minimal | Allows for long-term use without frequent intervention |
By focusing on these aspects, researchers can select and maintain reference electrodes that provide reliable and accurate data, contributing to the success of their electrochemical studies.
Conditions of Use for Different Reference Electrodes
Classification Based on pH
When selecting a reference electrode for electrochemical experiments, the pH of the electrolyte is a critical factor. Different reference electrodes are specifically designed to function optimally under varying pH conditions, ensuring accurate and stable measurements.
Acidic Conditions
For environments with low pH, calomel electrodes are typically employed. These electrodes are reliable in acidic settings due to their stability and the fact that they do not react with acidic solutions. The calomel electrode consists of mercury in contact with a paste of mercury (I) chloride (calomel) and a potassium chloride solution, which provides a stable potential.

Neutral Conditions
In neutral pH environments, silver chloride electrodes are the preferred choice. This type of electrode is composed of a silver wire coated with silver chloride, which is immersed in a potassium chloride or sodium chloride solution. The silver chloride electrode offers excellent stability and low temperature coefficient, making it suitable for a wide range of applications.
Alkaline Conditions
For high pH environments, mercury-mercury oxide electrodes are used. These electrodes are made of mercury in contact with a paste of mercury oxide and potassium hydroxide solution. The mercury-mercury oxide electrode is stable in alkaline conditions and provides a consistent potential, which is crucial for accurate measurements in such environments.
| Electrode Type | Optimal pH Range | Components |
|---|---|---|
| Calomel Electrode | Acidic (low pH) | Mercury, mercury (I) chloride paste, potassium chloride solution |
| Silver Chloride Electrode | Neutral (mid pH) | Silver wire coated with silver chloride, potassium chloride or sodium chloride solution |
| Mercury-Mercury Oxide Electrode | Alkaline (high pH) | Mercury, mercury oxide paste, potassium hydroxide solution |
Understanding the specific pH requirements for each type of reference electrode ensures that the most appropriate electrode is chosen, thereby enhancing the accuracy and reliability of electrochemical measurements.
Special Cases and Organic Electrolytes
In specific scenarios, such as those involving sulfuric acid or sulfate solutions, mercury mercuric sulfate electrodes are employed due to their compatibility with these harsh environments. These electrodes are particularly effective in maintaining stable potentials and ensuring accurate measurements in such aggressive conditions.
In the realm of battery technology, cadmium hydroxide electrodes are prevalent. These electrodes are chosen for their ability to withstand the cyclic demands of battery operations, providing reliable performance over extended periods.
For applications involving organic electrolytes, silver and ferrocene electrodes are often the preferred choices. Silver electrodes are valued for their stability and low reactivity with organic compounds, while ferrocene electrodes offer unique advantages in terms of their redox properties and compatibility with organic media.
| Electrolyte Type | Preferred Electrode | Key Advantages |
|---|---|---|
| Sulfuric Acid Solutions | Mercury Mercuric Sulfate | Compatibility with aggressive environments, stable potential |
| Battery Industry | Cadmium Hydroxide | Endurance in cyclic battery operations, reliable performance |
| Organic Electrolytes | Silver and Ferrocene | Stability with organic compounds, unique redox properties |
This table summarizes the preferred electrodes for different electrolyte types, highlighting their key advantages in various applications.
Why Different Reference Electrodes Apply to Different Conditions
Electrode Reaction and Nernst Equation
Different electrodes are tailored for specific conditions based on their unique electrode reactions and the resulting standard equilibrium electrode potentials, which are meticulously calculated using the Nernst equation. This equation, named after the German chemist Walther Nernst, is fundamental in electrochemistry as it relates the reduction potential of an electrochemical reaction to the standard electrode potential and the activities of the reacting species.
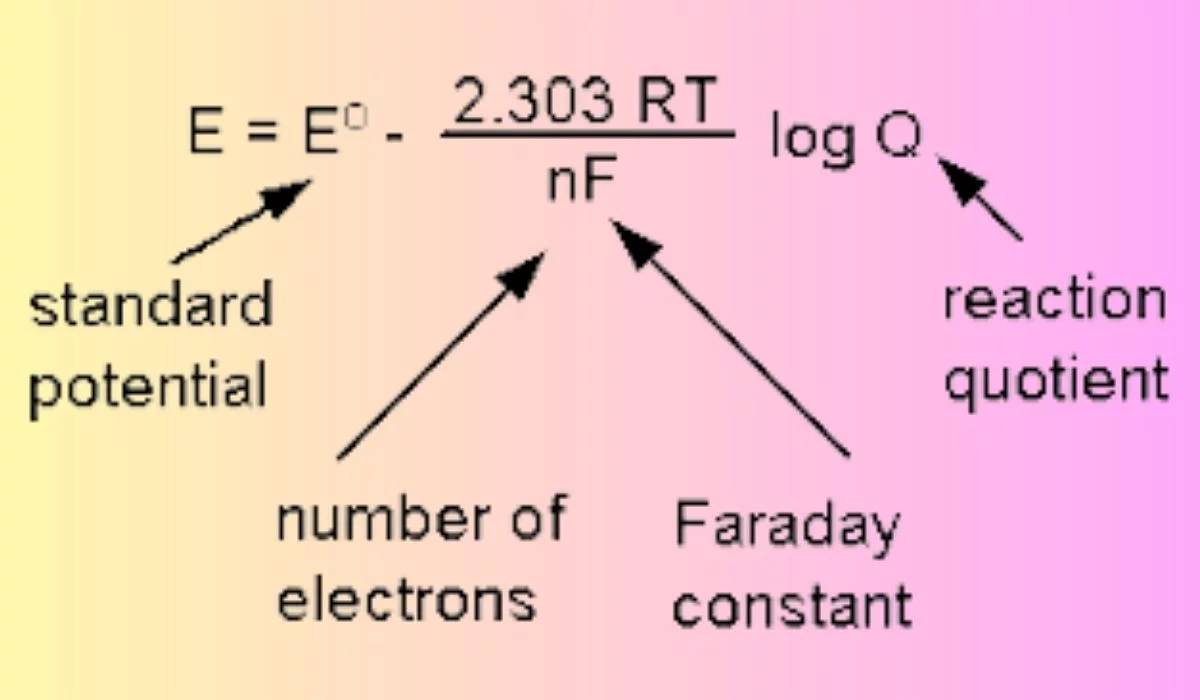
For instance, the standard equilibrium electrode potential of a silver chloride electrode, often used in neutral environments, is influenced by the concentration of chloride ions and the partial pressure of hydrogen gas. In contrast, a calomel electrode, favored in acidic conditions, operates under different thermodynamic principles, ensuring stability and accuracy in its designated environment.
| Electrode Type | Suitable Conditions | Key Reaction | Standard Equilibrium Potential (V) |
|---|---|---|---|
| Silver Chloride | Neutral | AgCl + e⁻ → Ag + Cl⁻ | 0.2223 |
| Calomel | Acidic | Hg₂Cl₂ + 2e⁻ → 2Hg + 2Cl⁻ | 0.2682 |
| Mercury-Mercury Oxide | Alkaline | HgO + H₂O + 2e⁻ → Hg + 2OH⁻ | 0.0977 |
The selection of an appropriate electrode is crucial as it directly impacts the accuracy and reliability of electrochemical measurements. Each electrode's suitability is determined by its ability to maintain a stable potential under varying conditions, ensuring that the Nernst equation accurately predicts the electrode's behavior. This precision is essential for applications ranging from environmental monitoring to battery technology, where even minor deviations can lead to significant errors.
In summary, the Nernst equation not only quantifies the relationship between electrode potential and reaction conditions but also underscores the importance of choosing the right electrode for specific applications. This ensures that electrochemical experiments yield precise and reproducible results, aligning with the broader objectives of scientific inquiry and industrial practice.
Stability and Disadvantages
The silver chloride electrode serves as a prime example of how specific conditions can impact the stability and accuracy of reference electrodes. Under alkaline conditions, prolonged use of this electrode can result in the formation of silver oxide. This reaction not only compromises the electrode's stability but also diminishes its accuracy, thereby affecting the reliability of the measurements taken.
In alkaline environments, the chemical reaction can be summarized as follows:
- Silver Chloride (AgCl) + Hydroxide (OH⁻) → Silver Oxide (Ag₂O) + Chloride (Cl⁻)
This transformation significantly alters the electrode's potential, making it less predictable and more prone to error. The instability introduced by the formation of silver oxide necessitates careful consideration when selecting and using reference electrodes in such conditions.
To mitigate these disadvantages, employing a double salt bridge system is often recommended. This method helps in maintaining the stability of the silver chloride electrode in alkaline conditions, ensuring more accurate and reliable readings.
Mitigating Disadvantages with Salt Bridges
In alkaline conditions, silver chloride electrodes can face stability issues due to the formation of silver oxide over time, which affects the accuracy and reliability of measurements. To counteract these disadvantages, employing a double salt bridge system is an effective strategy.
A double salt bridge system involves the use of two separate salt bridges, each filled with an appropriate electrolyte. This setup helps to isolate the reference electrode from the test solution, thereby reducing the risk of contamination and minimizing the interaction that could lead to the formation of silver oxide. By creating a buffer zone, the double salt bridge system ensures that the reference electrode remains stable, even in challenging alkaline environments.
This method not only preserves the integrity of the silver chloride electrode but also enhances the overall accuracy of electrochemical measurements. The dual isolation provided by the salt bridges acts as a protective barrier, allowing for more consistent and reliable data collection in alkaline conditions.
Related Products
- Reference Electrode Calomel Silver Chloride Mercury Sulfate for Laboratory Use
- Copper Sulfate Reference Electrode for Laboratory Use
- Metal Disc Electrode Electrochemical Electrode
- Graphite Disc Rod and Sheet Electrode Electrochemical Graphite Electrode
- Glassy Carbon Electrochemical Electrode



















