
Electrochemical Consumables
Rotating Platinum Disk Electrode for Electrochemical Applications
Item Number : ELEP
Price varies based on specs and customizations
$49.90 / set
- Specifications
- 0.5 ~ 6mm, can be customized
- Applicable temperature range
- 0 ~ 60℃
- Rod Material
- PTFE
- Guide material
- high Purity Platinum> 99.99%
Shipping:
Contact us to get shipping details Enjoy On-time Dispatch Guarantee.
Why Choose Us
Easy ordering process, quality products, and dedicated support for your business success.
Introduction
Disk electrodes are essential components for electrochemical experiments, commonly used in three-electrode setups. They are circular with a small ring around the edge and can be micromachined to a very small size. Disk electrodes are often used for cost-efficient disposable electrodes and are key applications for electrochemical techniques like cyclic voltammetry and impedance spectroscopy.
Technical specifications

| Specifications | 0.5 ~ 6mm, can be customized |
| Applicable temperature range | 0 ~ 60℃ |
| Rod Material | PTFE |
| Guide material | high Purity Platinum> 99.99% |
Detail & Parts



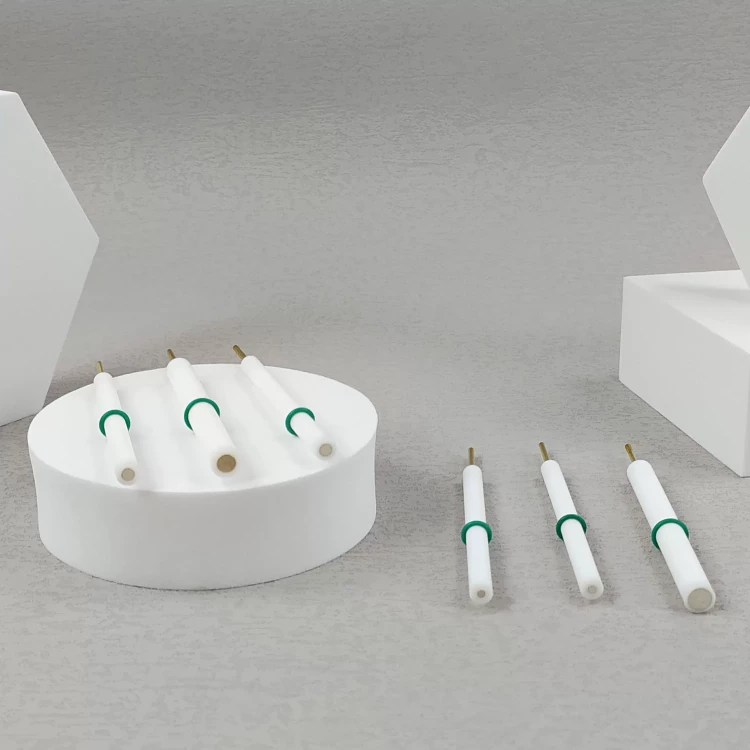



Applications
Platinum Disc Electrodes are widely used in various electrochemical techniques for substrate analysis. Here are some key application areas:
- Electrochemical analysis: Disk electrodes are used in a variety of electrochemical analysis techniques, such as cyclic voltammetry, chronoamperometry, and impedance spectroscopy. These techniques can be used to study the electrochemical properties of materials, such as their redox behavior, conductivity, and capacitance.
- Electrocatalytic measurements: Platinum Disc Electrodes are commonly used as the disk electrode in rotating ring-disk electrodes (RRDEs) for electrocatalytic measurements. Due to its low electrocatalytic activity compared to Pt and Au, glassy carbon (GC) is often used as the disk electrode onto which the electrocatalyst is deposited.
- Direct dropping experiments: Very small Platinum Disc Electrodes can be used for direct dropping experiments, where a small drop of a solution of interest is put directly onto the electrode and the electrical impedance is measured.
- Fuel cells: Platinum thin films are crucial in fuel cells, providing high electro-catalytic property with low resistance.
- Solar cells: Platinum counter electrodes are used in solar cells made of dye-synthesized TiO2, improving the ionic diffusion rate and enabling high current draw.
- Gas electrodes and polymers: Platinum is deposited on various substrates like gas electrodes and polymers through impregnation and reduction methods. The sputtering technique allows for precise distribution of metal and homogenous particles with controlled thickness.
Designed for You
KinTek provide deep custom made service and equipment to worldwide customers, our specialized teamwork and rich experienced engineers are capable to undertake the custom tailoring hardware and software equipment requirements, and help our customer to build up the exclusive and personalized equipment and solution!
Would you please drop your ideas to us, our engineers are ready for you now!
Trusted by Industry Leaders

FAQ
What Is An Electrode In Electrochemistry?
What Is Rotating Disk Electrode Used For?
What Is The Function Of Auxiliary Electrode?
What Are The Materials Used In Electrochemical Cell?
What Are The 3 Electrodes In Electrochemistry?
What Is The Rotating Electrode Method?
What Is The Difference Between Auxiliary And Reference Electrode?
What Are The Examples Of Electrochemical Material?
What Are The Different Types Of Electrochemical Electrodes?
What Is The Rotating Ring-disk Electrode Method?
What Materials Are Commonly Used For Auxiliary Electrodes?
What Materials Are Commonly Used For Electrochemical Electrodes?
What Are The Advantages Of Rotating Disc Electrode?
How Do Auxiliary Electrodes Affect The Performance Of An Electrochemical Cell?
What Factors Should Be Considered When Selecting An Electrochemical Electrode?
Why Are Auxiliary Electrodes Necessary In Electrochemical Systems?
How Can Electrochemical Electrodes Be Used In Various Applications?
Are There Any Limitations Or Considerations When Using Auxiliary Electrodes?
REQUEST A QUOTE
Our professional team will reply to you within one business day. Please feel free to contact us!
Related Products

RRDE rotating disk (ring disk) electrode / compatible with PINE, Japanese ALS, Swiss Metrohm glassy carbon platinum
Elevate your electrochemical research with our Rotating Disk and Ring Electrodes. Corrosion resistant and customizable to your specific needs, with complete specifications.

Platinum Sheet Electrode for Laboratory and Industrial Applications
Elevate your experiments with our Platinum Sheet Electrode. Crafted with quality materials, our safe and durable models can be tailored to fit your needs.

Platinum Sheet Electrode for Battery Lab Applications
Platinum sheet is composed of platinum, which is also one of the refractory metals. It is soft and can be forged, rolled and drawn into rod, wire, plate, tube and wire.

Platinum Auxiliary Electrode for Laboratory Use
Optimize your electrochemical experiments with our Platinum Auxiliary Electrode. Our high-quality, customizable models are safe and durable. Upgrade today!

Gold Disc Electrode
Looking for a high-quality gold disc electrode for your electrochemical experiments? Look no further than our top-of-the-line product.

Metal Disc Electrode Electrochemical Electrode
Elevate your experiments with our Metal Disk Electrode. High-quality, acid and alkali resistant, and customizable to fit your specific needs. Discover our complete models today.

Graphite Disc Rod and Sheet Electrode Electrochemical Graphite Electrode
High-quality graphite electrodes for electrochemical experiments. Complete models with acid and alkali resistance, safety, durability, and customization options.

High Purity Gold Platinum Copper Iron Metal Sheets
Elevate your experiments with our high-purity sheet metal. Gold, platinum, copper, iron, and more. Perfect for electrochemistry and other fields.

Gold Electrochemical Sheet Electrode Gold Electrode
Discover high-quality gold sheet electrodes for safe and durable electrochemical experiments. Choose from complete models or customize to meet your specific needs.

Glassy Carbon Electrochemical Electrode
Upgrade your experiments with our Glassy Carbon Electrode. Safe, durable, and customizable to fit your specific needs. Discover our complete models today.

Laboratory Disc Rotary Mixer for Efficient Sample Mixing and Homogenization
Efficient Laboratory Disc Rotary Mixer for Precise Sample Mixing, Versatile for Various Applications, DC Motor and Microcomputer Control, Adjustable Speed and Angle.

Reference Electrode Calomel Silver Chloride Mercury Sulfate for Laboratory Use
Find high-quality reference electrodes for electrochemical experiments with complete specifications. Our models offer resistance to acid and alkali, durability, and safety, with customization options available to meet your specific needs.

Laboratory CVD Boron Doped Diamond Materials
CVD boron-doped diamond: A versatile material enabling tailored electrical conductivity, optical transparency, and exceptional thermal properties for applications in electronics, optics, sensing, and quantum technologies.

Inclined Rotary Plasma Enhanced Chemical Vapor Deposition PECVD Equipment Tube Furnace Machine
Upgrade your coating process with PECVD coating equipment. Ideal for LED, power semiconductors, MEMS and more. Deposits high-quality solid films at low temps.

Electrode Polishing Material for Electrochemical Experiments
Looking for a way to polish your electrodes for electrochemical experiments? Our polishing materials are here to help! Follow our easy instructions for best results.

Copper Sulfate Reference Electrode for Laboratory Use
Looking for a Copper Sulfate Reference Electrode? Our complete models are made of high-quality materials, ensuring durability and safety. Customization options available.

Electrode Fixture for Electrochemical Experiments
Upgrade your experiments with our customizable Electrode Fixtures. High-quality materials, acid and alkali resistant, and safe and durable. Discover our complete models today.

Electric Rotary Kiln Pyrolysis Furnace Plant Machine Calciner Small Rotary Kiln Rotating Furnace
Electric rotary kiln - precisely controlled, it's ideal for calcination and drying of materials like lithium cobalate, rare earths, and non-ferrous metals.
Related Articles

Comprehensive Guide to Rotating Disk Electrode (RDE) in Electrochemical Studies
Explore the detailed workings, applications, and significance of Rotating Disk Electrodes (RDE) in electrochemical research. Discover how RDEs are used in fuel cells, catalyst development, and more.

Understanding the Rotating Disk Electrode: Principles and Applications
Explores the development, principles, and applications of the rotating disk electrode in electrochemistry.

Basic Principles and Applications of Rotating Disc Electrodes
Explores the development, principles, and applications of Rotating Disc Electrodes in electrochemistry.

Introduction to Rotating Disc Electrodes and Common Electrochemical Applications
An overview of rotating disc electrodes and their applications in various electrochemical studies, including catalyst evaluation, battery research, and corrosion protection.

Exploring Rotating Electrode Technology in Electrochemistry
An in-depth analysis of rotating electrode technology, its applications, and its impact on electrochemical reactions under different flow conditions.

Advantages of the Rotating Electrode Method
Rotating electrode method involves rotating a small working electrode to increase the mass transport of reactants and products, allowing for more precise measurements and better control of reaction conditions.

Electrochemical Electrodes in Chemical Analysis
Electrochemical electrodes are essential tools used in many chemical analysis techniques and experiments. These electrodes are devices that allow us to measure the electrical potential difference in a chemical reaction.

Electrolytes and Electrochemical Electrodes
Electrolytes and electrodes play an essential role in electrochemistry. Electrolytes are substances that conduct electricity when dissolved in water or melted.

The Future of Electrochemical Electrodes
The latest trends and developments in electrode materials and their implications for the future of electrochemistry.

Innovations in Electrochemical Electrodes Technology
Recent advancements in nanotechnology and materials science have led to significant improvements in electrochemical devices, making them more efficient, durable, and cost-effective.

Understanding Electrodeposition with Electrochemical Electrodes
Electrodeposition is a process of depositing a metal or a non-metallic material onto a surface by applying an electric current.

How to Choose the Right Electrochemical Electrode
The choice of electrode material can have a significant impact on the performance of the electrochemical system.