
Electrochemical Consumables
Platinum Auxiliary Electrode for Laboratory Use
Item Number : ELPA
Price varies based on specs and customizations
$25.90 / set
- Features
- Corrosion Resistant
- Applicable temperature range
- 0 ~ 80℃
- Wire diameter
- 0.5 / 1mm
Shipping:
Contact us to get shipping details Enjoy On-time Dispatch Guarantee.
Why Choose Us
Easy ordering process, quality products, and dedicated support for your business success.
Introduction
Auxiliary electrodes are often fabricated from electrochemically inert materials such as gold, platinum, or carbon. The material used to make an auxiliary electrode must be an inert material like graphite or a noble metal such as gold, carbon, or platinum. The purpose of the auxiliary electrode is to provide a pathway for current to flow in the electrochemical cell without passing significant current through the reference electrode.
The Platinum Auxiliary Electrode is ideal for electrochemical experiments, featuring complete models constructed with high-quality materials. It is not only safe and durable, but also customizable to meet your specific needs.
Technical specifications
Platinum Wire Ring Electrode

| Features | Corrosion Resistant |
| Applicable temperature range | 0 ~ 80℃ |
| Wire diameter | 0.5 / 1mm |
| Material | pure platinum |
| Customize material (gold, silver, platinum, copper) and rod length to your needs. | |
Platinum wire electrode

| Features | Corrosion Resistant |
| Applicable temperature range | 0 ~ 80℃ |
| Wire diameter | 0.5 / 1mm |
| Material | PTFE Rod + Platinum Wire |
| Customize material (gold, silver, platinum, copper) and rod length to your needs. | |
60 mesh platinum mesh electrode

| Features | Corrosion Resistant |
| Applicable temperature range | 0 ~ 80℃ |
| Size | 5*5~ 50*50mm |
| Material | PTFE Rod + Platinum Mesh |
| Pure platinum wire braided mesh electrode, high purity and large surface area. | |
Platinum column electrode
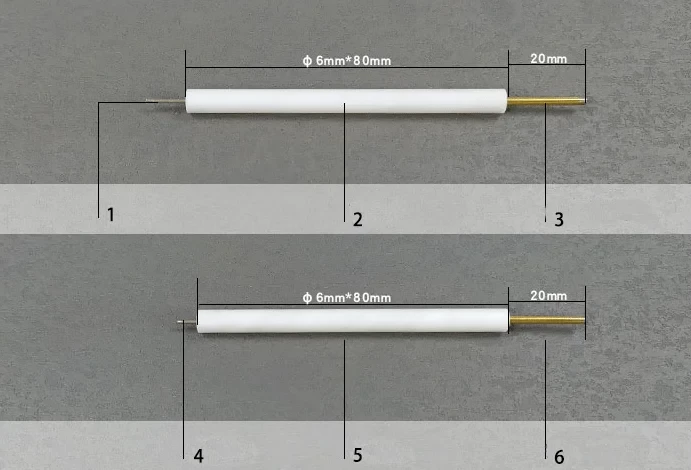
| Features | Corrosion Resistant |
| Applicable temperature range | 0 ~ 80℃ |
| Wire diameter | 1mm ~ 2mm |
| Material | PTFE Rod + Platinum |
| Customize material (gold, silver, platinum, copper) and rod length to your needs. | |
Detail & Parts










Features
The Platinum Auxiliary Electrode is an essential component for any electrochemistry setup. It provides a stable and inert surface for the transfer of electrons between the working electrode and the solution, ensuring accurate and reliable measurements.
- High surface area: The platinum gauze working electrode has a high surface area, which allows for a greater current flow and faster electron transfer, making it ideal for bulk electrolysis.
- Inert material: Platinum is an inert material, which means that it will not react with the solution or the working electrode, ensuring that the measurements are not affected by any chemical reactions.
- Corrosion-resistant: Platinum is a corrosion-resistant metal, which means that it will not corrode or degrade over time, ensuring a long lifespan for the electrode.
- Durable: Platinum is a durable material, which means that it can withstand repeated use and harsh conditions, making it an excellent choice for demanding applications.
- Versatile: The Platinum Auxiliary Electrode can be used in a wide range of applications, including electrochemistry, electroplating, and corrosion testing, making it a valuable tool for any laboratory.
Applications
Platinum auxiliary electrodes are widely used in various electrochemical applications, including:
- Electrochemical cells: As the counter electrode in electrochemical cells, facilitating electron transfer and maintaining charge balance.
- Voltammetry: As the counter electrode in cyclic voltammetry (CV) experiments, allowing for the study of electrochemical processes and determination of redox potentials.
- Electrolysis: As the counter electrode in electrolysis experiments, providing electrons for reduction reactions and ensuring efficient current flow.
- Fuel cells: As the counter electrode in fuel cells, facilitating electron transfer and enhancing cell efficiency.
- Solar cells: As the counter electrode in dye-synthesized TiO2 solar cells, improving ionic diffusion and enabling higher current output.
- Sputtering: As the target material in sputtering processes, producing thin platinum films with high electro-catalytic properties and low resistance.
Designed for You
KinTek provide deep custom made service and equipment to worldwide customers, our specialized teamwork and rich experienced engineers are capable to undertake the custom tailoring hardware and software equipment requirements, and help our customer to build up the exclusive and personalized equipment and solution!
Would you please drop your ideas to us, our engineers are ready for you now!
Trusted by Industry Leaders

FAQ
What Is The Purpose Of A Platinum Auxiliary Electrode?
What Are The Benefits Of Using A Platinum Auxiliary Electrode?
How Do I Choose The Right Platinum Auxiliary Electrode?
What Is An Electrode In Electrochemistry?
What Is The Function Of Auxiliary Electrode?
What Are The 3 Electrodes In Electrochemistry?
What Is The Difference Between Auxiliary And Reference Electrode?
What Are The Different Types Of Electrochemical Electrodes?
What Materials Are Commonly Used For Auxiliary Electrodes?
What Materials Are Commonly Used For Electrochemical Electrodes?
How Do Auxiliary Electrodes Affect The Performance Of An Electrochemical Cell?
What Factors Should Be Considered When Selecting An Electrochemical Electrode?
Why Are Auxiliary Electrodes Necessary In Electrochemical Systems?
How Can Electrochemical Electrodes Be Used In Various Applications?
Are There Any Limitations Or Considerations When Using Auxiliary Electrodes?
REQUEST A QUOTE
Our professional team will reply to you within one business day. Please feel free to contact us!
Related Products

Platinum Sheet Electrode for Laboratory and Industrial Applications
Elevate your experiments with our Platinum Sheet Electrode. Crafted with quality materials, our safe and durable models can be tailored to fit your needs.

Platinum Sheet Electrode for Battery Lab Applications
Platinum sheet is composed of platinum, which is also one of the refractory metals. It is soft and can be forged, rolled and drawn into rod, wire, plate, tube and wire.

Rotating Platinum Disk Electrode for Electrochemical Applications
Upgrade your electrochemical experiments with our Platinum Disc Electrode. High-quality and reliable for accurate results.

RRDE rotating disk (ring disk) electrode / compatible with PINE, Japanese ALS, Swiss Metrohm glassy carbon platinum
Elevate your electrochemical research with our Rotating Disk and Ring Electrodes. Corrosion resistant and customizable to your specific needs, with complete specifications.

High Purity Gold Platinum Copper Iron Metal Sheets
Elevate your experiments with our high-purity sheet metal. Gold, platinum, copper, iron, and more. Perfect for electrochemistry and other fields.

Gold Disc Electrode
Looking for a high-quality gold disc electrode for your electrochemical experiments? Look no further than our top-of-the-line product.

Gold Electrochemical Sheet Electrode Gold Electrode
Discover high-quality gold sheet electrodes for safe and durable electrochemical experiments. Choose from complete models or customize to meet your specific needs.

Reference Electrode Calomel Silver Chloride Mercury Sulfate for Laboratory Use
Find high-quality reference electrodes for electrochemical experiments with complete specifications. Our models offer resistance to acid and alkali, durability, and safety, with customization options available to meet your specific needs.

Graphite Disc Rod and Sheet Electrode Electrochemical Graphite Electrode
High-quality graphite electrodes for electrochemical experiments. Complete models with acid and alkali resistance, safety, durability, and customization options.

Metal Disc Electrode Electrochemical Electrode
Elevate your experiments with our Metal Disk Electrode. High-quality, acid and alkali resistant, and customizable to fit your specific needs. Discover our complete models today.

Glassy Carbon Electrochemical Electrode
Upgrade your experiments with our Glassy Carbon Electrode. Safe, durable, and customizable to fit your specific needs. Discover our complete models today.

Laboratory CVD Boron Doped Diamond Materials
CVD boron-doped diamond: A versatile material enabling tailored electrical conductivity, optical transparency, and exceptional thermal properties for applications in electronics, optics, sensing, and quantum technologies.

CF KF Flange Vacuum Electrode Feedthrough Lead Sealing Assembly for Vacuum Systems
Discover high-vacuum CF/KF flange electrode feedthroughs, ideal for vacuum systems. Superior sealing, excellent conductivity, and customizable options.

Copper Sulfate Reference Electrode for Laboratory Use
Looking for a Copper Sulfate Reference Electrode? Our complete models are made of high-quality materials, ensuring durability and safety. Customization options available.

Electrode Polishing Material for Electrochemical Experiments
Looking for a way to polish your electrodes for electrochemical experiments? Our polishing materials are here to help! Follow our easy instructions for best results.

Conductive Carbon Cloth Carbon Paper Carbon Felt for Electrodes and Batteries
Conductive carbon cloth, paper, and felt for electrochemical experiments. High-quality materials for reliable and accurate results. Order now for customization options.

Nickel Aluminum Tabs for Soft Pack Lithium Batteries
Nickel tabs are used to manufacture cylindrical and pouch batteries, and positive aluminum and negative nickel are used to produce lithium-ion and nickel batteries.

H Type Electrolytic Cell Triple Electrochemical Cell
Experience versatile electrochemical performance with our H-type Electrolytic Cell. Choose from membrane or non-membrane sealing, 2-3 hybrid configurations. Learn more now.

Electrode Fixture for Electrochemical Experiments
Upgrade your experiments with our customizable Electrode Fixtures. High-quality materials, acid and alkali resistant, and safe and durable. Discover our complete models today.

Li-Air Battery Case for Battery Lab Applications
Lithium air battery (lithium oxygen battery) dedicated battery box. The positive electrode is punched from the inside out, and the inside is smooth.
Related Articles

Types of Bias Power Supplies in Magnetron Sputtering and Their Purposes
Overview of bias power supply types in magnetron sputtering and their roles in improving film adhesion and density.

The Invisible Architecture of Accuracy: Mastering Electrode Installation
Master the lifecycle of electrode installation—from inspection to alignment and maintenance—to ensure safety and reproducibility in electrochemical experiments.

The Silent Interface: Mastery Over Electrode Decay
Electrode failure is rarely sudden; it is the compound interest of neglect. Learn the disciplined maintenance protocols that preserve accuracy and longevity.

Understanding Electrodes and Electrochemical Cells
An electrode is a point where current enters and leaves the electrolyte. It is a conductor used to make a junction with a nonmetallic part of a circuit. Electrodes can be made of materials such as gold, platinum, carbon, graphite, or metal. They serve as the surface for oxidation-reduction reactions in electrochemical cells. There are different types of electrodes, including anode and cathode.

Understanding Electrodes in Electrochemical Systems: Working, Counter, and Reference Electrodes
An overview of the roles and characteristics of working, counter, and reference electrodes in electrochemical systems.

Electrochemistry Basics: Conditions and Precautions for Using Different Reference Electrodes
A guide on the requirements and conditions for using various reference electrodes in electrochemistry.

A Beginner's Guide to Understanding Reference Electrodes in Electrochemistry
Reference electrodes provide a stable and known potential that other electrodes can be compared to, allowing for accurate measurements of electrochemical reactions.

Common Reference Electrodes in Electrochemical Studies
An overview of the key requirements and types of reference electrodes used in electrochemical systems, particularly in lithium-ion batteries.

Reference Electrodes: Calomel, Silver Chloride, and Mercury Sulfate - A Comprehensive Guide
Explore the world of reference electrodes, including calomel, silver chloride, and mercury sulfate. Understand their construction, principles, and applications in electrochemical measurements.

Pseudo-Reference Electrodes When and How to Use Them
Reference electrodes are an essential component in electrochemical measurements. They are used to establish a stable and reproducible potential for the working electrode and provide a reference point for the measurement. The reference electrode should have a stable and well-defined potential, which is independent of the solution's composition and the working electrode's potential.

How to Choose the Right Reference Electrode for Your Application
When it comes to measuring the electrical potential of a solution accurately, a reference electrode is an essential tool in the laboratory. It provides a stable and consistent reference point for other electrodes to measure against, ensuring accurate and reliable results.

A Guide to Choosing the Right Reference Electrode for Your ISE Analysis
Reference electrodes are an essential component of any electrochemical measurement.