The Invisible Variable
In laboratory science, we often obsess over the invisible. We worry about electron transfer rates, impedance, and the purity of reagents measured in parts per million.
But we rarely stop to admire the vessel that holds it all together.
This is an oversight. In electrochemistry, the quartz cell is not merely a container; it is a boundary condition. It defines the limits of what is possible. The difference between a failed experiment and a breakthrough often lies in the architecture of the cell itself—specifically, the number and size of the holes drilled into its lid.
At KINTEK, we see these specifications not just as dimensions, but as a map of experimental intent.
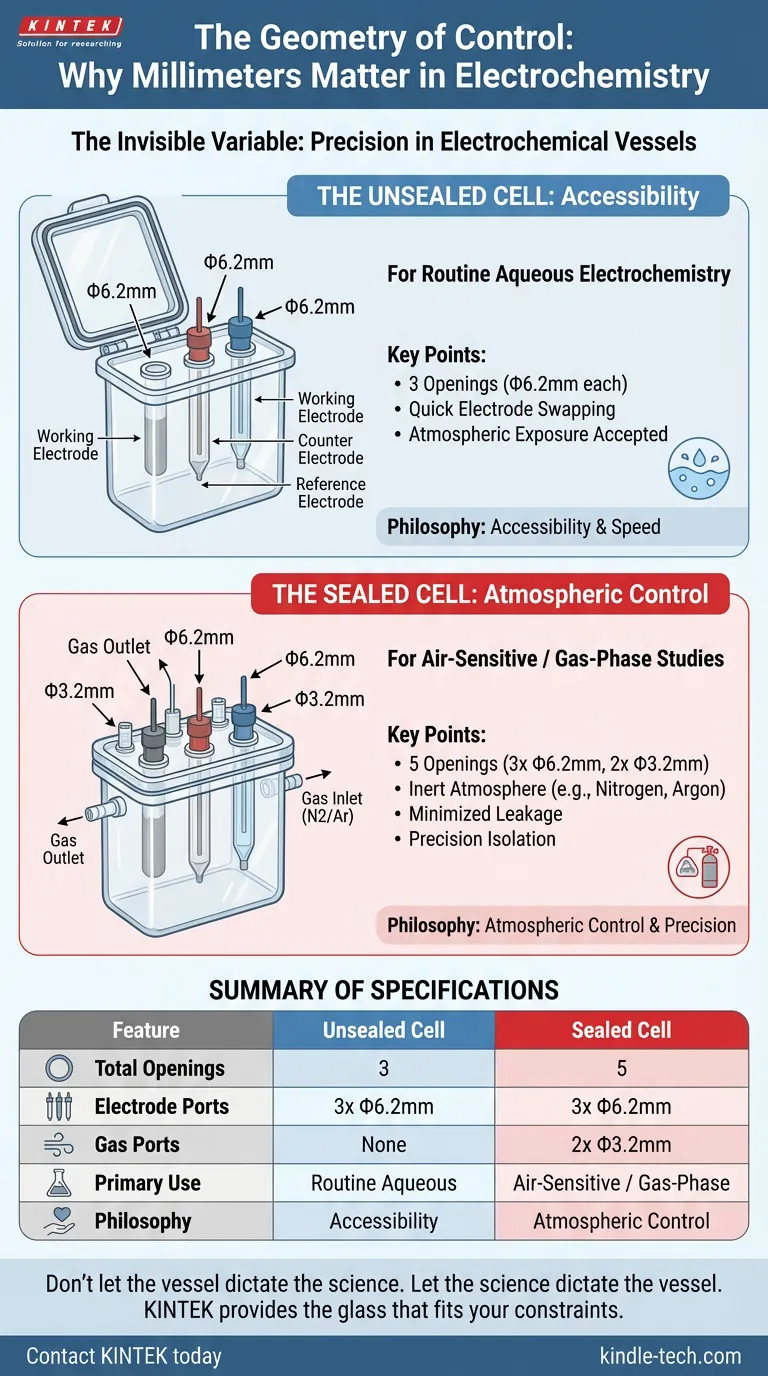
The Philosophy of the Unsealed Cell
The standard unsealed all-quartz electrolytic cell represents the science of the routine. It is designed for environments where the atmosphere is benign.
The architecture is deceptively simple. It features three openings.
Each opening has a standardized diameter of Φ6.2mm.
This number is not arbitrary. It is the physical manifestation of the classic three-electrode system:
- The Working Electrode
- The Counter Electrode
- The Reference Electrode
The unsealed cell is a wager. You are betting that the ambient air—the oxygen, the humidity—will not interfere with your chemistry. For general aqueous solutions, this is a safe bet. The large, uniform openings allow for quick electrode swapping and easy cleaning. It prioritizes speed and accessibility.
The Architecture of Isolation
However, chemistry is rarely compliant.
When you move into the realm of air-sensitive materials or organic solvents, the atmosphere becomes a contaminant. Precision requires isolation. This is where the sealed cell becomes necessary.
A sealed cell changes the geometry of the experiment. It increases the port count to five.
It retains the foundation of the unsealed version:
- Three openings at Φ6.2mm (for the standard electrodes).
But it adds a critical layer of control:
- Two openings at Φ3.2mm.
These smaller ports are the gatekeepers. They are dedicated to gas inlets and outlets. They allow you to purge the headspace with nitrogen or argon, creating an inert blanket over your reaction.
The difference in diameter—6.2mm versus 3.2mm—is a functional distinction. The larger ports accommodate the bulk of the electrode bodies, while the smaller ports are sized for tight tubing connections, minimizing the surface area for potential leaks.
The Cost of Standardization
Standardization makes science scalable. Knowing that a "standard" cell will fit your electrodes allows you to focus on the data, not the hardware.
However, there is a hidden cost to standardization: rigidity.
Real-world research often drifts outside the margins of "standard." You might need a Luggin capillary to minimize IR drop. You might need to insert a temperature probe or a pH sensor.
In these moments, the standard 3-port or 5-port configuration becomes a limitation.
This is why the best equipment suppliers treat standards as a starting point, not a conclusion. While the Φ6.2mm/Φ3.2mm setup covers 90% of use cases, the remaining 10%—often the most innovative work—requires customization.
Summary of Specifications
To select the right vessel, you must first define the enemy. Is it time (efficiency needed) or the atmosphere (control needed)?
| Feature | Unsealed Cell | Sealed Cell |
|---|---|---|
| Total Openings | 3 | 5 |
| Electrode Ports | 3x Φ6.2mm | 3x Φ6.2mm |
| Gas Ports | None | 2x Φ3.2mm |
| Primary Use | Routine aqueous electrochemistry | Air-sensitive / Gas-phase studies |
| Philosophy | Accessibility | Atmospheric Control |
Choosing Your Constraints
Every piece of lab equipment enforces a set of constraints.
An unsealed cell constrains you to stable environments. A sealed cell constrains you to a more complex setup process but grants you freedom from atmospheric interference.
At KINTEK, we provide the glass that fits your specific set of constraints. Whether you need the rugged simplicity of a standard 3-port cell or a custom-designed array for a complex multi-sensor experiment, the goal remains the same: reliable, reproducible data.
Don't let the vessel dictate the science. Let the science dictate the vessel.
Visual Guide
Related Products
- Quartz Electrolytic Electrochemical Cell for Electrochemical Experiments
- Electrolytic Electrochemical Cell with Five-Port
- PTFE Electrolytic Cell Electrochemical Cell Corrosion-Resistant Sealed and Non-Sealed
- Electrolytic Electrochemical Cell for Coating Evaluation
- Flat Corrosion Electrolytic Electrochemical Cell
Related Articles
- The Art of the Empty Vessel: Preparing Quartz Electrolytic Cells for Absolute Precision
- Electrochemistry The Science Behind Electrochemical Cells
- The Architecture of Silence: Why Quartz Defines Electrochemical Precision
- Benefits of Electrochemical Cells for Energy Storage
- The Architecture of Transparency: Mastering Safety and Precision in Quartz Electrolysis




















