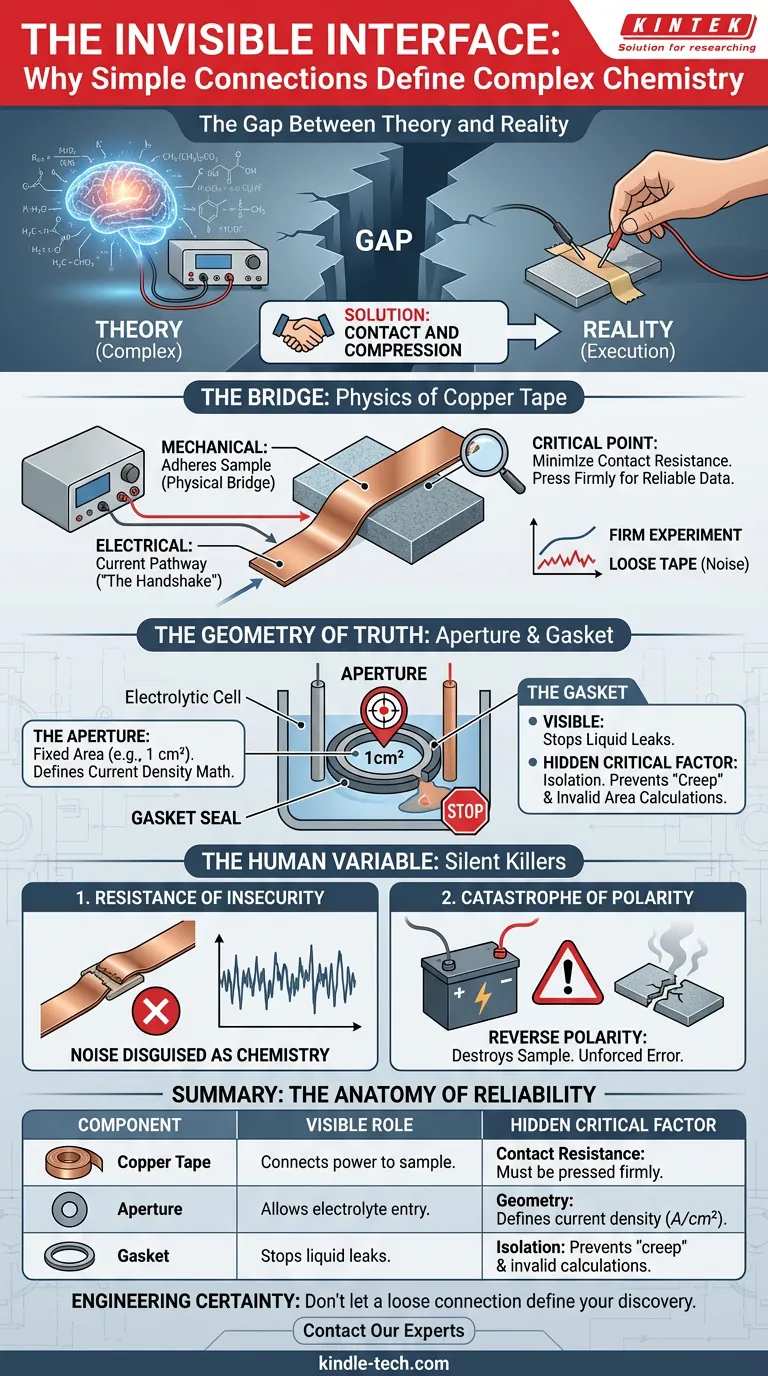
The Gap Between Theory and Reality
We tend to romanticize the complex parts of science. In electrochemistry, we obsess over the Nernst equation, the sophisticated potentiostat, or the molecular structure of the electrolyte.
We assume that if the theory is sound, the result is inevitable.
But the most common point of failure in a laboratory isn't usually a flaw in the chemical theory. It is a flaw in the physical execution. It is the gap between the macroscopic world (the wire) and the microscopic world (the sample).
Bridging that gap requires a solution that feels almost too simple to be scientific: contact and compression.
The Bridge: The Physics of Copper Tape
To make a working electrode sample conductive, you don't need a complex laser weld. You need a reliable physical bridge. In this electrolytic cell design, that bridge is copper tape.
It serves a dual purpose that appeals to the engineer’s desire for efficiency:
- Mechanical: It physically adheres the sample to the cell’s contact.
- Electrical: It creates the pathway for current to flow from the power supply to the material.
This is the "handshake" of the experiment. If the copper tape is loose, the handshake is weak. Resistance spikes. The voltage drop becomes unpredictable.
The data you see on your screen is only as honest as the piece of tape holding your sample.
The Geometry of Truth
Once the connection is made, the experiment needs boundaries. In science, undefined variables are the enemy of reproducibility.
The cell utilizes a strict physical design to impose order on the chaotic behavior of ions:
- The Aperture: A fixed circular hole, typically 1 cm².
- The Gasket: A seal that isolates the reaction.
Why the Gasket Matters
The gasket does more than prevent leaks. It defines the "truth" of your data.
Current density is calculated based on surface area. If the electrolyte leaks under the gasket, your reactive surface area increases effectively unknown to you. Your calculations for current density will be wrong, not because the chemistry failed, but because the geometry failed.
The gasket ensures that only the exposed portion participates in the reaction. It forces the universe of your experiment to exist within that defined 1 cm² circle.
The Human Variable
Atul Gawande often writes that the volume of knowledge in medicine has exceeded our ability to manage it safely without checklists. The same applies to the lab bench.
The machinery is perfect; the operator is human. There are two "silent killers" of electrochemical data:
1. The Resistance of Insecurity
An insecure physical connection—where the copper tape isn't pressed firmly—introduces contact resistance. This noise disguises itself as chemical behavior. You might spend weeks analyzing a "peak" in your data that is actually just a loose piece of tape.
2. The Catastrophe of Polarity
The most dangerous mistake is the simplest: Reverse Polarity.
Connecting positive to negative incorrectly isn't just a data error. It can trigger unintended reactions that destroy the sample or damage the electrode. It is an unforced error that happens when we are rushed.
Summary: The Anatomy of Reliability
Understanding the system allows you to troubleshoot the result.
| Component | The Visible Role | The "Hidden" Critical Factor |
|---|---|---|
| Copper Tape | Connects power to sample | Contact Resistance: Must be pressed firmly to minimize noise. |
| Aperture | Allows electrolyte entry | Geometry: Defines the math behind current density ($A/cm^2$). |
| Gasket | Stops liquid leaks | Isolation: Prevents "creep" that invalidates surface area calculations. |
Engineering Certainty
Great science is about controlling variables. You cannot control the electron, but you can control the connection.
At KINTEK, we understand that the reliability of your data hangs by a thread—or in this case, a piece of copper tape and a gasket. We design our lab equipment and consumables to remove the guesswork from these physical interfaces, allowing you to focus on the chemistry rather than the mechanics.
Don't let a loose connection define your discovery.
Visual Guide
Related Products
- Custom PTFE Teflon Parts Manufacturer for Gaskets and More
- Button Battery Case Gasket for Battery Lab Applications
- Copper Foam
- Zirconia Ceramic Gasket Insulating Engineering Advanced Fine Ceramics
Related Articles
- Polytetrafluoroethylene (PTFE): How low friction coefficient promotes industrial progress
- PTFE gasket: an all-around solution for flange sealing
- How to use PTFE to improve the working efficiency of pumps and valves
- Performance and application of polytetrafluoroethylene (PTFE) in high temperature environment
- PTFE's high temperature and corrosion resistance: Why it is indispensable in industry




