The Invisible Vessel
In experimental science, we often fixate on the actors and ignore the stage.
We obsess over the purity of the catalyst. We agonize over the voltage parameters. We spend hours polishing the working electrode until it mirrors our own exhaustion. But we rarely give a second thought to the glass vessel holding it all together.
This is a mistake. In electrochemistry, the cell is not just a container; it is a boundary condition.
The standard five-port water bath electrolytic cell is the workhorse of modern electrochemistry. It represents a precise attempt to impose order on a chaotic chemical environment. But to use it effectively, you must understand the logic behind its architecture—and when that logic fails to meet your specific needs.
Deconstructing the "Standard"
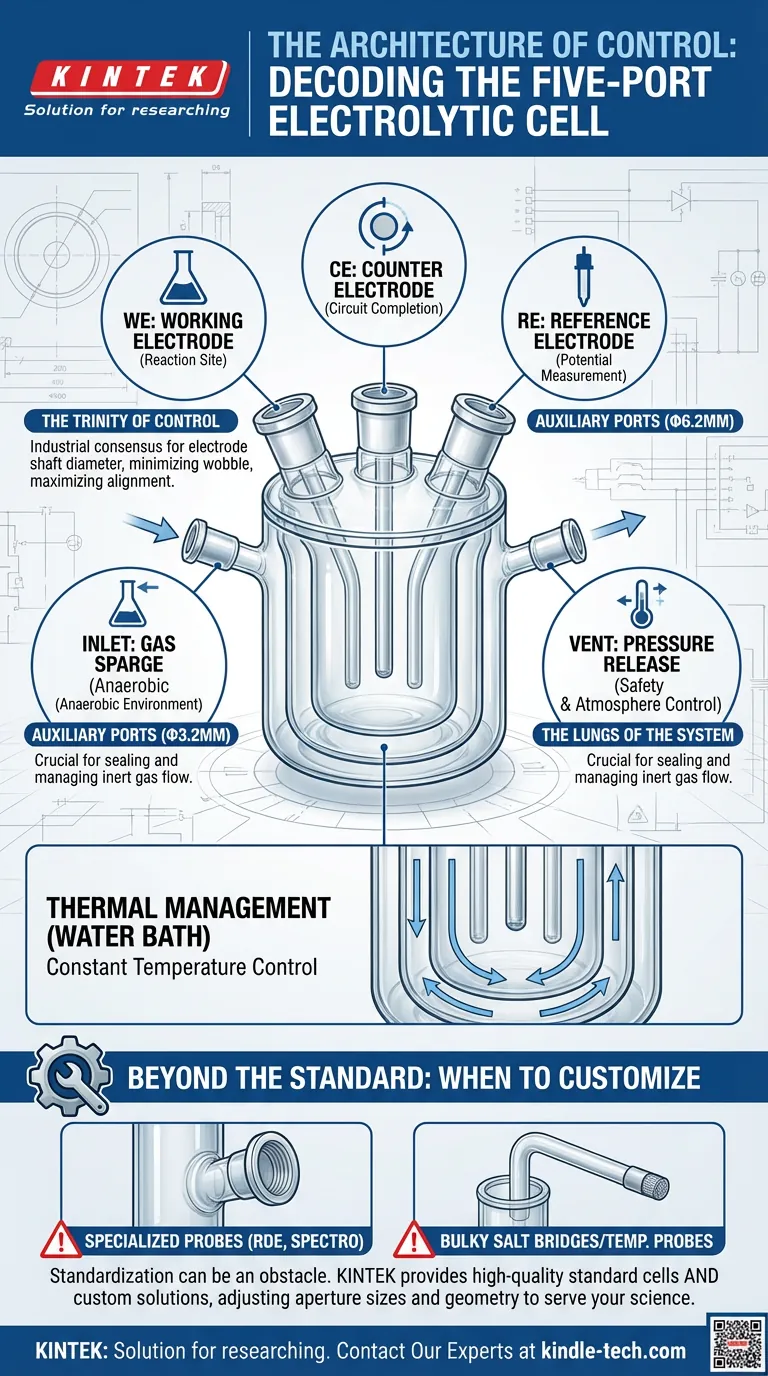
When manufacturers speak of a "standard" sealed cell, they are referring to a specific geometric legacy designed to accommodate the classic three-electrode system.
The configuration is almost always a 3+2 arrangement. It is a system built for governance: three ports to control the reaction, and two ports to control the atmosphere.
The Trinity of Control (Φ6.2mm)
The three largest apertures typically feature a diameter of Φ6.2mm. These are the structural pillars of your experiment.
- The Working Electrode (WE): The site of the reaction you are studying.
- The Counter Electrode (CE): The component that completes the circuit.
- The Reference Electrode (RE): The stable yardstick against which potential is measured.
Why 6.2mm? It is not an arbitrary number. It is the industrial consensus for electrode shaft diameter, allowing for a snug fit that minimizes wobble and maximizes alignment.
The Lungs of the System (Φ3.2mm)
The two smaller apertures, usually Φ3.2mm, handle the gas management.
Electrochemistry often demands an anaerobic environment. Oxygen is an aggressive interferent. To combat this, one port acts as the inlet for inert gases (like Nitrogen or Argon) to purge the solution. The second port serves as the vent, preventing pressure build-up that could shatter the glass or compromise seals.
The Psychological Trap of "Standard"
There is a psychological comfort in buying "standard" equipment. It suggests that the path has been beaten for you, that the variable has been solved.
But in research, "standard" is merely a baseline, not a universal law.
A standard configuration assumes you are running a standard experiment. It assumes your reference electrode is a standard size. It assumes you do not need a temperature probe submerged in the electrolyte. It assumes you are not using a Rotating Disk Electrode (RDE), which requires a significantly larger central port.
If you treat the standard specification as rigid, you force your experiment to compromise. You end up using adapters that leak, or tilting electrodes at angles that skew current distribution.
The Engineering of the Interface
The quality of your data is often determined by the quality of your seals.
The five-port design is most critical in sealed cell systems. If the apertures (Φ6.2mm and Φ3.2mm) do not perfectly match your PTFE stoppers or electrode shafts, the atmosphere is compromised.
Furthermore, the "water bath" aspect introduces a second layer of complexity: thermal management. The double-jacketed design allows a heat transfer fluid to circulate around the reaction. This turns the cell into a thermostat, locking in temperature as a constant rather than a variable.
Summary of Specifications
Here is the baseline logic for the standard configuration:
| Port Type | Quantity | Diameter | Function |
|---|---|---|---|
| Primary Ports | 3 | Φ6.2mm | Working, Counter, & Reference Electrodes |
| Auxiliary Ports | 2 | Φ3.2mm | Gas Inlet (Sparging) & Outlet (Venting) |
Choosing the Right Tool
Great engineering is about matching the tool to the constraint.
If you are performing general cyclic voltammetry in a controlled atmosphere, the standard 3x(Φ6.2mm) + 2x(Φ3.2mm) configuration is likely the elegant solution you need. It is time-tested and robust.
However, if your research pushes the edges—using specialized spectroelectrochemical probes, RDEs, or bulky salt bridges—standardization becomes an obstacle. In these cases, the "standard" is the wrong tool. You need customization.
The KINTEK Approach
At KINTEK, we appreciate the romance of the perfect setup. We understand that a glass cell is a precision instrument, not a jar.
We provide high-quality standard cells for routine excellence, but we also recognize that innovation often requires breaking the mold. Whether you need to adjust aperture sizes, add ports for temperature monitoring, or redesign the geometry for a custom reactor, we ensure the glass serves your science, not the other way around.
Visual Guide
Related Products
- Electrolytic Electrochemical Cell with Five-Port
- Double Layer Five-Port Water Bath Electrolytic Electrochemical Cell
- Customizable PEM Electrolysis Cells for Diverse Research Applications
- PTFE Electrolytic Cell Electrochemical Cell Corrosion-Resistant Sealed and Non-Sealed
- Side Window Optical Electrolytic Electrochemical Cell
Related Articles
- The Architecture of Accuracy: Inside the Five-Port Electrolytic Cell
- The Transparency Paradox: Mastering the Fragile Art of Electrolytic Cells
- Electrolytic Batteries A Solution to the Growing Energy Demands
- The Symphony of Coefficients: Why Your Electrolytic Cell Cannot Be a Monolith
- Electrochemistry The Science Behind Electrochemical Cells




















