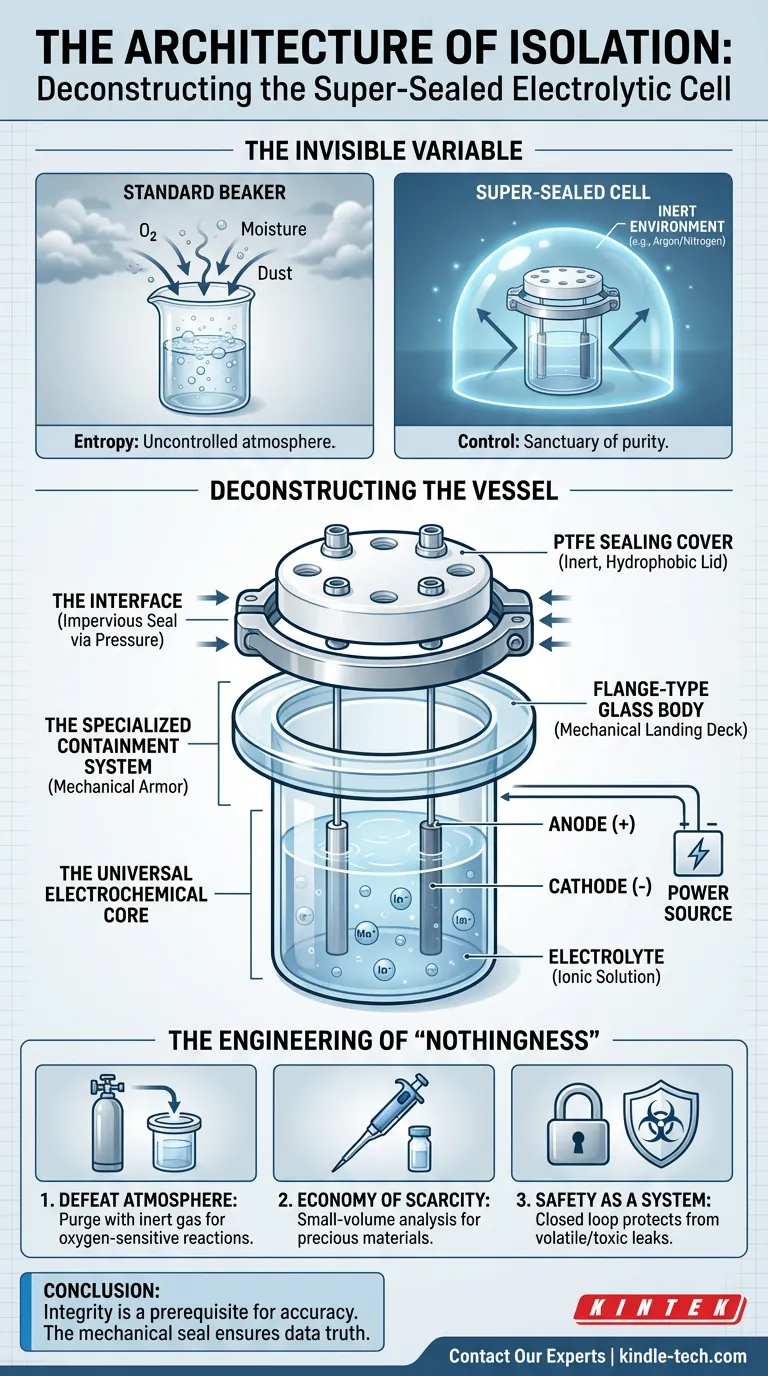
The Invisible Variable
In experimental science, we obsess over the visible variables. We measure voltage with precision. We calculate molarity to the third decimal point. We select electrode materials with agonizing care.
But often, the variable that ruins an experiment is the one you cannot see: the atmosphere.
A standard beaker is an open invitation to entropy. Oxygen creeps in. Moisture settles. For general chemistry, this is negligible. For sensitive electrochemical analysis, it is catastrophic.
This is the psychological gap that the super-sealed electrolytic cell bridges. It is not merely a container; it is a mechanism of control. It transforms the laboratory bench—a chaotic environment of dust and air—into a sanctuary of chemical purity.
To understand why this matters, we must look past the chemistry and appreciate the mechanical engineering that makes it possible.
Deconstructing the Vessel
At a glance, an electrolytic cell seems simple. It holds liquid. It holds metal. It conducts current. However, a super-sealed cell is defined by a specific division of labor between its chemical heart and its mechanical armor.
The Universal Electrochemical Core
Every cell, regardless of its sophistication, relies on three non-negotiable elements. These are the actors in the play:
- The Electrodes: The anode and cathode. These are the conductive bridges that allow energy to enter the system.
- The Electrolyte: The ionic solution. This is the medium that completes the circuit.
The Specialized Containment System
The "super-sealed" designation does not change the chemistry; it changes the stage. It introduces a high-integrity housing system designed to hermetically seal the actors away from the audience.
- The Flange-Type Glass Body: Unlike a smooth-rimmed beaker, this vessel ends in a flange—a wide, flat, protruding lip. This is not an aesthetic choice; it is a mechanical landing deck.
- The PTFE Sealing Cover: The lid is crafted from Polytetrafluoroethylene (Teflon). It is chemically inert, hydrophobic, and tough.
- The Interface: When the PTFE cover is clamped against the glass flange, it creates a seal that is impervious to gas exchange.
The Engineering of "Nothingness"
Why go to such lengths to trap a liquid?
The value of a super-sealed cell lies in its ability to maintain "nothingness." By excluding the outside world, you ensure that the only reactions occurring are the ones you initiated.
1. Defeating the Atmosphere
Many advanced electrochemical reactions are oxygen-sensitive. The presence of air can oxidize the sample or skew the current response. The flange-and-clamp design allows researchers to purge the cell with inert gases like argon or nitrogen, creating a controlled vacuum or inert environment that holds for the duration of the experiment.
2. The Economy of Scarcity
Not all electrolytes are essentially saltwater. Some researchers work with rare isotopes or expensive, synthesized catalysts.
A super-sealed cell often features a compact geometry. Because the seal prevents evaporation and spills, it allows for small-volume analysis. You can run rigorous experiments with micro-liters of solution, preserving precious resources that would otherwise be lost to the walls of a larger vessel or evaporation.
3. Safety as a System
If your electrolyte is volatile, toxic, or corrosive, a leak is not just a failed experiment; it is a safety hazard. The mechanical pressure of the flange seal converts the cell into a closed loop, protecting the operator from the chemistry.
The Trade-Off: Complexity vs. Necessity
Complexity is a cost. In engineering, you never pay for complexity unless it buys you reliability.
The super-sealed cell is more expensive to manufacture than a standard cell. It requires precision machining of the PTFE and high-quality glass blowing for the flange. It takes longer to assemble. It is harder to clean.
When should you pay this cost?
- The Standard Cell: If you are demonstrating electrolysis for a class or working with stable, aqueous solutions, the super-sealed design is overkill. The complexity impedes the workflow.
- The Super-Sealed Cell: If your results depend on the absence of oxygen, or if your materials cost more than the glassware itself, the standard cell is a liability.
Summary of Architecture
Understanding the physical structure helps in selecting the right tool.
| Component | Material | The "Why" |
|---|---|---|
| Cell Body | Flange-Type Glass | Provides a flat, machinable surface for a high-pressure seal. |
| Lid | PTFE (Teflon™) | Resists chemical attack; effectively "gaskets" against the glass. |
| Mechanism | Clamps/Screws | Applies the physical force necessary to merge the lid and body. |
| Environment | Isolated | Prevents ingress of O2/H2O; prevents egress of volatiles. |
Conclusion
The super-sealed electrolytic cell is a testament to a simple truth in science: Integrity is a prerequisite for accuracy.
You cannot analyze what you cannot contain. While the chemistry provides the data, it is the mechanical seal—the boring, static marriage of glass and plastic—that ensures the data is true.
At KINTEK, we understand that your equipment is the foundation of your discovery. Whether you are dealing with highly volatile electrolytes or conducting air-sensitive research, our specialized lab equipment is engineered to eliminate the variables you didn't plan for.
Contact Our Experts today to discuss how our super-sealed cells can bring absolute certainty to your electrochemical experiments.
Visual Guide
Related Products
- Super Sealed Electrolytic Electrochemical Cell
- PTFE Electrolytic Cell Electrochemical Cell Corrosion-Resistant Sealed and Non-Sealed
- Double-Layer Water Bath Electrolytic Electrochemical Cell
- Double Layer Five-Port Water Bath Electrolytic Electrochemical Cell
- Electrolytic Electrochemical Cell for Coating Evaluation
Related Articles
- The Transparency Paradox: Mastering the Fragile Art of Electrolytic Cells
- The Art of Isolation: Why Super-Sealed Cells Define Modern Electrochemistry
- The Geometry of Control: Inside the Super-Sealed Electrolytic Cell
- The Art of the Non-Spontaneous: Precision in Electrolytic Circuits
- Electrolytic Batteries A Solution to the Growing Energy Demands



















