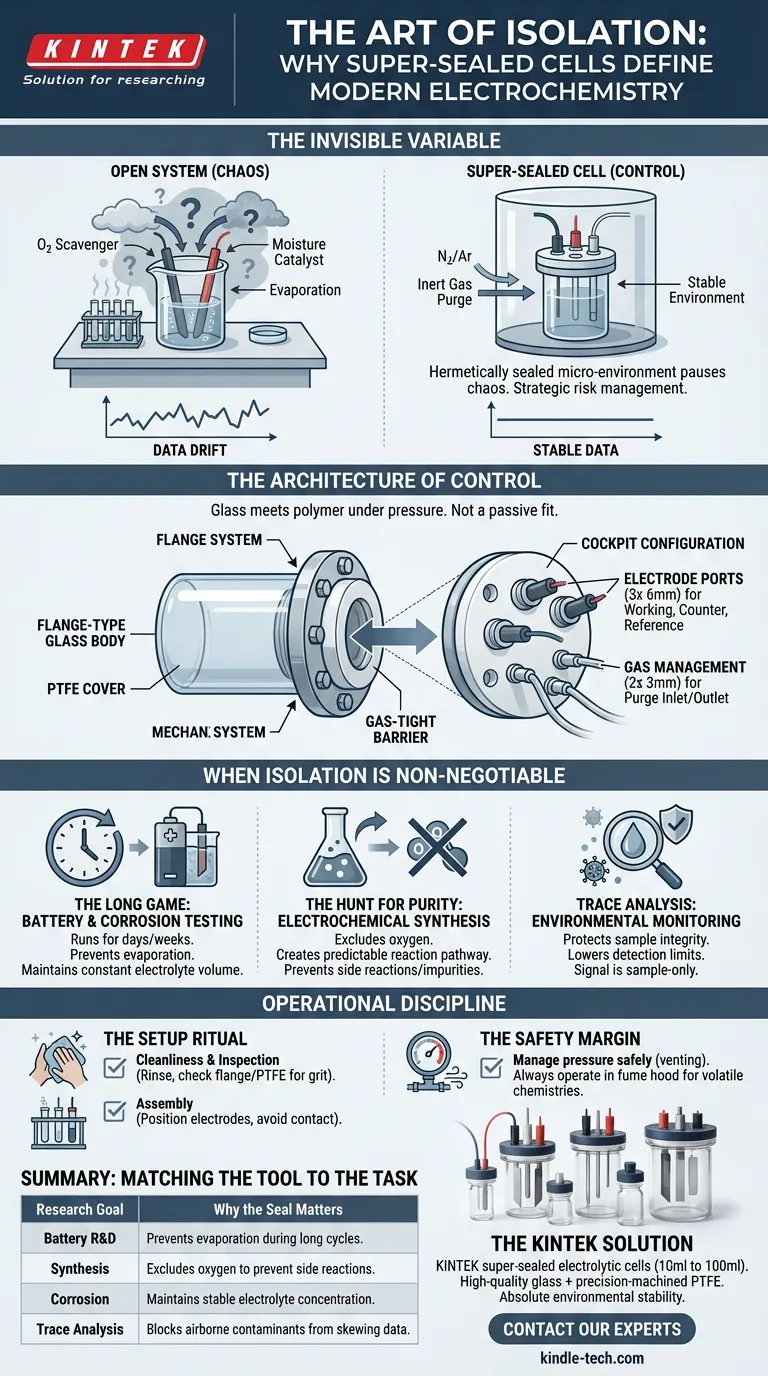
The Invisible Variable
In scientific research, the most dangerous variable is often the one you assume is constant.
In electrochemistry, we obsess over voltage, current, and electrode material. We calculate potentials to the millivolt. Yet, the integrity of the data is frequently undermined by something much simpler: the atmosphere.
Oxygen is a scavenger. Moisture is a catalyst. Even the slow, silent evaporation of a solvent over a 24-hour cycle changes concentration levels, rendering data drift indistinguishable from actual reaction kinetics.
This is why the super-sealed electrolytic cell is not merely a container. It is a strategic tool for risk management.
By creating a hermetically sealed micro-environment, these cells allow researchers to pause the chaos of the outside world and focus entirely on the reaction at hand.
The Architecture of Control
To understand the value of a super-sealed cell, one must appreciate the engineering behind the seal. Standard glassware relies on gravity or loose friction fits. That is insufficient for high-stakes chemistry.
The super-sealed cell utilizes a flange-type glass body mated to a Polytetrafluoroethylene (PTFE) cover.
This is not a passive fit. The components are mechanically pressed together via a flange system. The result is an interface where glass meets polymer under pressure, creating a barrier that gases cannot cross.
The "Cockpit" Configuration
The design is strictly utilitarian, built for the three-electrode system. The PTFE cover acts as the command center, typically featuring:
- Electrode Ports: Three 6mm openings for the working, counter, and reference electrodes.
- Gas Management: Two 3mm openings for purging tubes (inlet and outlet).
This layout allows the researcher to introduce an inert gas (like Argon or Nitrogen) to displace oxygen, and then seal the system completely.
When Isolation is Non-Negotiable
Not every experiment requires this level of rigor. But for specific applications, the lack of a seal is a critical failure point.
The utility of the super-sealed cell becomes apparent in three distinct scenarios:
1. The Long Game: Battery and Corrosion Testing
Battery cycling and corrosion studies are battles of attrition. They run for hours, days, or weeks.
In an open system, the electrolyte evaporates. As the volume decreases, the concentration of salts increases. This shifts the conductivity and pH, introducing artificial trends into the data. A super-sealed cell maintains the volume constant from hour one to hour one hundred.
2. The Hunt for Purity: Electrochemical Synthesis
When synthesizing novel compounds, oxygen is often a competitor. It creates side reactions and impurities.
By mechanically locking out the atmosphere, the super-sealed cell ensures that the electrons go exactly where you intend them to go, creating a predictable reaction pathway.
3. Trace Analysis: Environmental Monitoring
When measuring pollutants at the parts-per-billion level, the lab air itself is a contaminant. The seal protects the sample integrity, lowering detection limits and ensuring that the signal you see comes from the sample, not the room.
Operational Discipline
A tool is only as effective as the hands that use it. The super-sealed cell requires a specific protocol to function correctly.
The Setup Ritual
- Cleanliness: The glass and electrodes must be rinsed with distilled water and appropriate solvents.
- Inspection: The flange and PTFE surface must be free of grit. A single particle can compromise the seal.
- Assembly: Electrodes must be positioned so they do not touch the glass walls or each other.
The Safety Margin Because the cell is sealed, it handles pressure differently than an open beaker. If a reaction generates significant gas, the purge lines must be managed correctly to vent pressure safely. Always operate within a fume hood when dealing with volatile or corrosive chemistries.
Summary: Matching the Tool to the Task
The choice of cell dictates the quality of the result.
| Research Goal | Why the Seal Matters |
|---|---|
| Battery R&D | Prevents evaporation during long charge/discharge cycles. |
| Synthesis | Excludes oxygen to prevent side reactions/impurities. |
| Corrosion | Maintains stable electrolyte concentration over days. |
| Trace Analysis | Blocks airborne contaminants from skewing data. |
The KINTEK Solution
At KINTEK, we understand that in the lab, control is everything.
Our super-sealed electrolytic cells—ranging from 10ml to 100ml—are designed for researchers who cannot afford to question their environmental baselines. By combining high-quality glass with precision-machined PTFE, we provide the isolation your chemistry demands.
If your research requires absolute environmental stability, it is time to upgrade your infrastructure.
Visual Guide
Related Products
- Super Sealed Electrolytic Electrochemical Cell
- PTFE Electrolytic Cell Electrochemical Cell Corrosion-Resistant Sealed and Non-Sealed
- Thin-Layer Spectral Electrolysis Electrochemical Cell
- Double-Layer Water Bath Electrolytic Electrochemical Cell
- Electrolytic Electrochemical Cell for Coating Evaluation
Related Articles
- The Transparency Paradox: Mastering the Fragile Art of Electrolytic Cells
- The Invisible Variable: Why Electrolytic Cell Hygiene Defines Data Integrity
- The Art of the Non-Spontaneous: Precision in Electrolytic Circuits
- The Architecture of Control: Mastering the Super-Sealed Electrolytic Cell
- The Silent Half of the Experiment: The Ritual of Cell Preservation



















