The Invisible Cost of Cleanliness
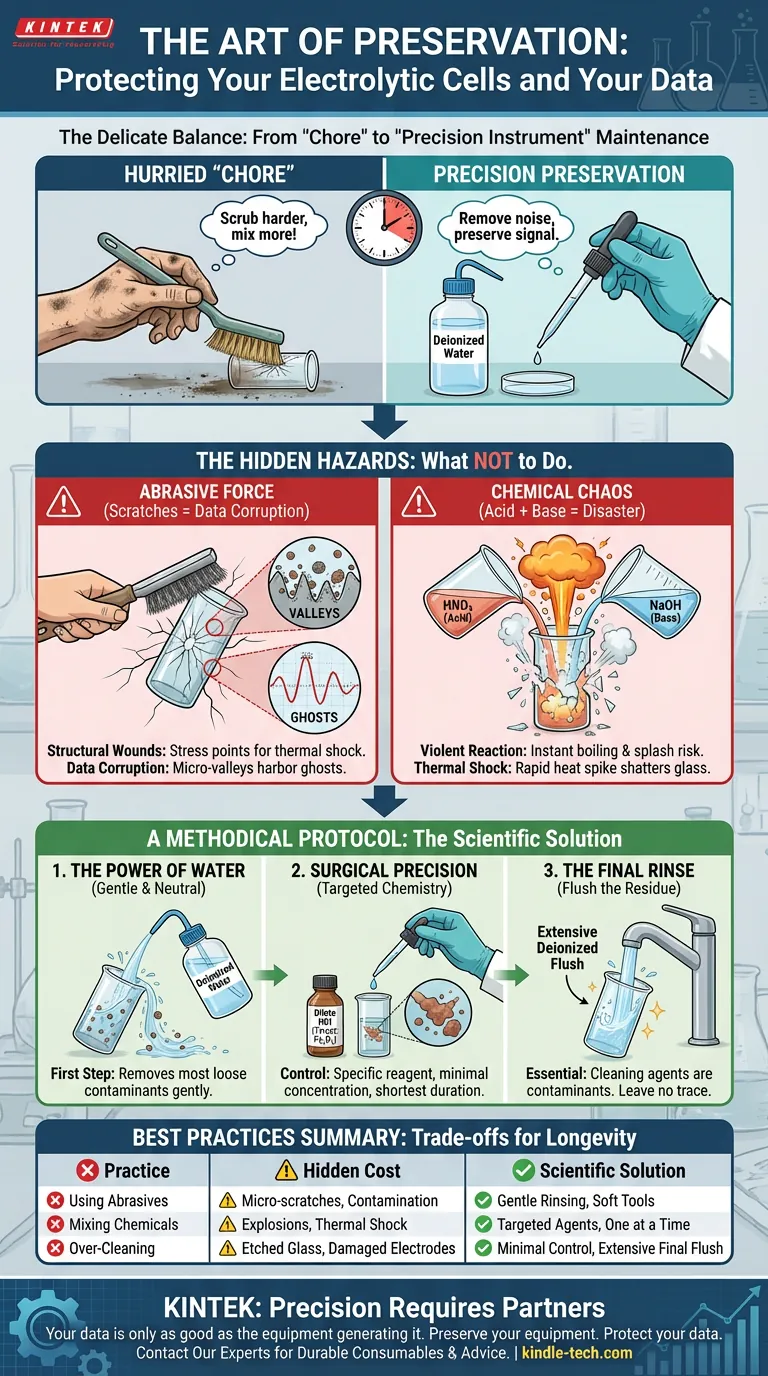
In the laboratory, we often treat cleaning as a chore—a hurried interval between the "real work" of experimentation.
This is a dangerous mindset.
The electrolytic cell is not merely a container; it is a precision instrument. It is the stage upon which your data is performed. Treating it like dirty dishes in a sink is the fastest way to compromise your results and destroy your investment.
The core principle of maintenance is preservation. Your goal is not just to remove contaminants. Your goal is to remove the noise without destroying the signal.
Here is how to navigate the delicate balance between hygiene and hazard.
The Psychology of Force (And Why You Must Resist It)
There is a human tendency to equate physical effort with cleanliness. If a stain is stubborn, our instinct is to scrub harder.
Do not do this.
The Problem with Abrasives
Using metal brushes or abrasive tools on an electrolytic cell is catastrophic. The body of the cell is typically glass—a material defined by its clarity and chemical inertness, but also its fragility.
Scratches are not cosmetic defects. They are structural wounds.
- Structural Integrity: A scratch is a stress concentrator, a weak point waiting for thermal shock to shatter the vessel.
- Data Corruption: Scratches create microscopic valleys where contaminants hide. These residues interfere with future electrochemical measurements, creating "ghosts" in your data.
The Chemical Fallacy
The second error stems from impatience. We assume that if one chemical is good, two must be better.
Never mix acid and base cleaning agents within the cell.
Combining nitric acid (HNO₃) and sodium hydroxide (NaOH) triggers a violent exothermic reaction. The solution boils instantly.
The result is a two-fold disaster:
- Personal Injury: You risk being splashed with boiling corrosive chemicals.
- Thermal Shock: The rapid heat spike can shatter the glass instantly.
A Methodical Protocol
Great science is boring. It is the result of repeatable, safe, and gentle processes. Cleaning your cell should be no different.
1. The Power of Water
Start with the simplest solvent. A thorough rinse with deionized water removes the vast majority of loose electrolytes and unreacted materials.
It is gentle, neutral, and safe. This should always be your first step.
2. Surgical Precision
If water fails, escalate to chemistry. But do so with the mindset of a surgeon, not a demolition crew.
- Target the Enemy: Use a reagent specific to the contaminant. Use dilute hydrochloric acid (HCl) for iron oxides. Do not use a "catch-all" cocktail.
- Control the Variables: Manage the concentration and the duration. Use the weakest effective solution for the shortest necessary time.
3. The Final Rinse
The cleaning agent is itself a contaminant.
Once the deposit is removed, you must flush the cell extensively with deionized water. If you leave cleaning residue behind, your next experiment is essentially testing the soap, not the sample.
The Trade-off: Efficiency vs. Longevity
Every maintenance decision involves a trade-off.
You can save five minutes by scrubbing with a wire brush. You can save ten minutes by mixing a high-potency chemical brew.
But these shortcuts compound. They etch the glass. They degrade the electrode surfaces. Eventually, they break the cell or invalidate a week's worth of data.
The smart engineer chooses the minimum effective intervention.
Summary of Best Practices
| Practice | The Hidden Cost | The Scientific Solution |
|---|---|---|
| Using Abrasives | Creates micro-scratches that harbor contaminants. | Gentle Rinsing: Use soft tools and deionized water. |
| Mixing Chemicals | Exothermic reactions cause explosions or thermal shock. | Targeted Agents: Use one specific chemical at a time. |
| Over-Cleaning | Etches glass and damages sensitive electrodes. | Control: Minimal concentration, minimal time. |
| Skipping Rinse | Residue ruins future experimental baselines. | Flush: Extensive final rinse with deionized water. |
Precision Requires Partners
Your data is only as good as the equipment generating it.
At KINTEK, we understand the "engineer's romance" with precision. We build our lab equipment and electrolytic cells to withstand the rigors of serious science, but they rely on your careful hand to last a lifetime.
Whether you need durable consumables or advice on the specific chemical compatibility of your cleaning agents, we are here to ensure your lab runs without interruption.
Preserve your equipment. Protect your data. Contact Our Experts to discuss the right tools for your laboratory.
Visual Guide
Related Products
- PTFE Electrolytic Cell Electrochemical Cell Corrosion-Resistant Sealed and Non-Sealed
- Super Sealed Electrolytic Electrochemical Cell
- Electrolytic Electrochemical Cell with Five-Port
- H Type Electrolytic Cell Triple Electrochemical Cell
- Electrolytic Electrochemical Cell Gas Diffusion Liquid Flow Reaction Cell
Related Articles
- Advanced Electrolytic Cell Techniques for Cutting-Edge Lab Research
- The Architecture of Invisibility: Deconstructing the "All-Quartz" Cell
- Electrolytic Cell Design and Standards
- Electrolytic Batteries A Solution to the Growing Energy Demands
- The Silent Partner: Why Material Choice in Electrochemistry is a Matter of Trust




















