In the intricate dance of electrochemistry, we often obsess over the visible actors: the catalyst, the electrolyte, and the applied potential. We meticulously polish electrodes and purge oxygen.
Yet, we frequently ignore the stage itself.
The electrolytic cell is often treated as a passive container—a simple bucket for ions. This is a dangerous oversimplification. The volume of your cell is not merely a capacity specification; it is a boundary condition that dictates the thermodynamics and kinetics of your experiment.
Whether you are working with a compact 10ml setup or a bulk 1000ml reactor, the geometry of the vessel defines the quality of the result.
The Architecture of a Reaction
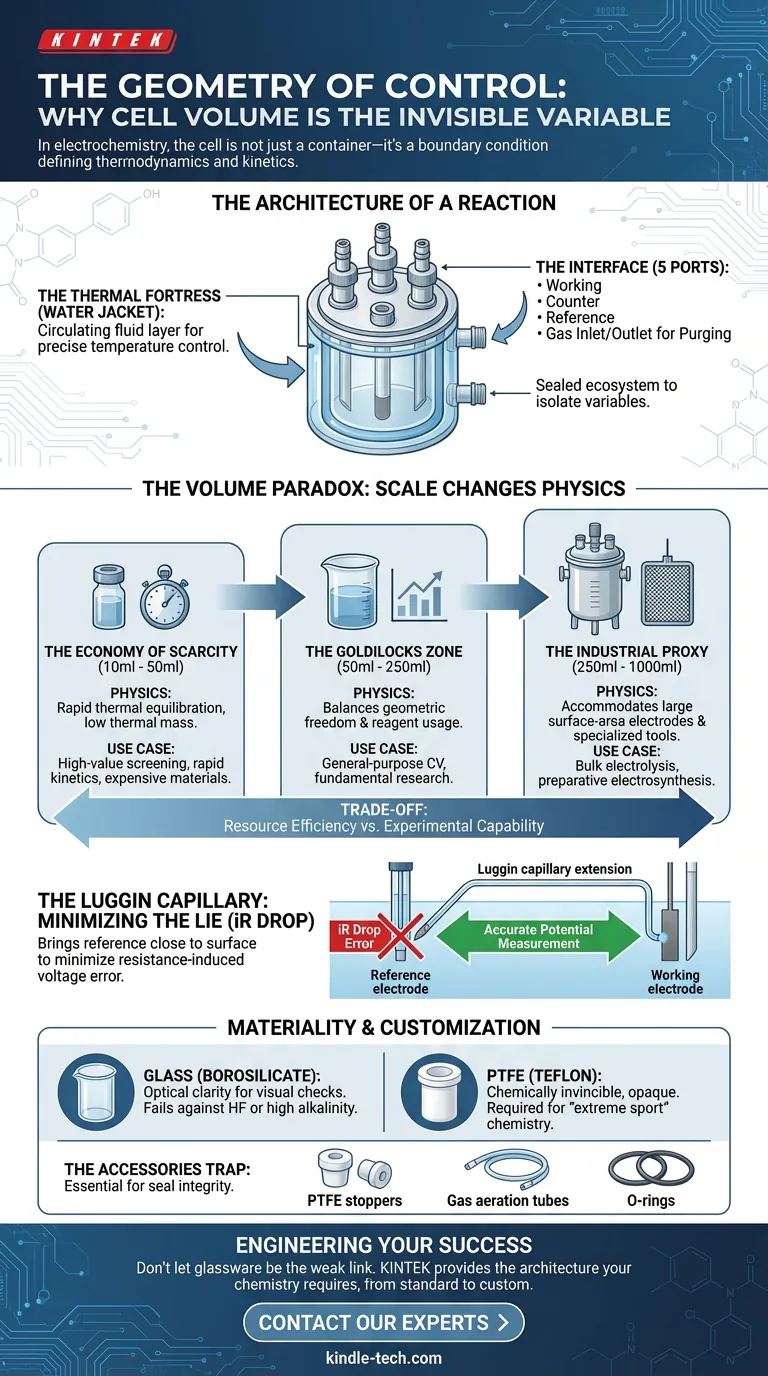
A standard five-port water bath electrolytic cell is not just glassware; it is a controlled environment designed to isolate variables. When you look at the design, you are looking at a system built to combat the entropy of the lab environment.
The Thermal Fortress (The Water Jacket) Temperature fluctuations are the enemy of reproducibility. A shift of a few degrees can drastically alter reaction rates and diffusion coefficients.
The double-walled "water jacket" design wraps your experiment in a circulating fluid layer. It acts as a thermal fortress, locking the internal environment to a precise setpoint, regardless of the ambient room temperature.
The Interface (The Ports) The "five-port" configuration is the standard interface between the macroscopic world (your instrumentation) and the microscopic world (the double layer).
- Three Primary Ports: For the working, counter, and reference electrodes.
- Two Auxiliary Ports: For gas inlet/outlet (purging).
This sealed ecosystem allows you to maintain an inert atmosphere, shielding sensitive chemistry from atmospheric oxygen.
The Volume Paradox: Scale Changes Physics
The typical volume range for these cells spans 10ml to 1000ml. While 50ml to 500ml is the commercial standard, the choice of volume forces a trade-off between resource efficiency and experimental capability.
You must choose the volume that matches the physics of your goal.
The Economy of Scarcity (10ml - 50ml)
In analytical chemistry, less is often more.
- The Constraint: You are working with rare catalysts or expensive deuterated solvents.
- The Physics: Small volumes have a lower thermal mass. They equilibrate with the water jacket almost instantly.
- The Use Case: High-value screening and rapid kinetic studies.
The Goldilocks Zone (50ml - 250ml)
This is the workhorse range found in most academic and industrial labs.
- The Constraint: Balancing reagent usage with geometric freedom.
- The Physics: Large enough to prevent rapid pH drift during electrolysis, but small enough to handle easily on a benchtop.
- The Use Case: General-purpose cyclic voltammetry (CV) and fundamental research.
The Industrial Proxy (250ml - 1000ml)
When you move to synthesis, the rules change.
- The Constraint: You need product, not just data.
- The Physics: These cells accommodate large surface-area electrodes (like large platinum mesh) and specialized tools like Rotating Disk Electrodes (RDE).
- The Use Case: Bulk electrolysis and preparative electrosynthesis.
The Luggin Capillary: Minimizing the Lie
One of the most critical components of the five-port design is the Luggin capillary.
In any electrochemical measurement, resistance in the solution creates an error in the voltage reading—the iR drop. This is effectively a "lie" told by the resistance of the electrolyte.
The Luggin capillary is a glass extension that brings the reference electrode tip within millimeters of the working electrode. It doesn't eliminate resistance, but it minimizes the distance the sensing element has to look through. It ensures that the potential you measure is the potential that actually exists at the surface.
Materiality and Customization
We tend to assume "standard" means "universal." It does not.
Glass vs. PTFE While borosilicate glass offers optical clarity—essential for checking bubble formation or electrode positioning—it fails against hydrofluoric acid or highly alkaline solutions. In these "extreme sport" chemistry scenarios, an all-PTFE (Teflon) body is required. It is opaque, but chemically invincible.
The Accessories Trap A cell body without the right fittings is a paperweight. The integrity of the seal relies on:
- PTFE stoppers machined to fit the ports exactly.
- Gas aeration tubes for proper sparging.
- Correct O-rings to prevent gas leaks.
Summary: Matching Geometry to Purpose
To choose a cell is to choose your constraints. Use the following guide to align your equipment with your experimental reality:
| Volume Range | Primary Physics | Ideal Application |
|---|---|---|
| 10ml - 50ml | Rapid Thermal Equilibration | Precious samples, expensive solvents, analytical screening. |
| 50ml - 250ml | Geometric Flexibility | Standard three-electrode experiments, cyclic voltammetry. |
| 250ml - 1000ml | High Capacity | Bulk electrolysis, synthesis, large electrode surface areas. |
Engineering Your Success
The difference between a failed experiment and a breakthrough often lies in the invisible variables. Don't let your choice of glassware be the weak link in your chain of evidence.
KINTEK understands that "off-the-shelf" doesn't always fit the unique demands of advanced research. Whether you need a standard 50ml glass cell or a custom PTFE reactor for harsh environments, we provide the architecture your chemistry requires.
Contact Our Experts today to discuss how we can configure the perfect electrolytic environment for your research.
Visual Guide
Related Products
- PTFE Electrolytic Cell Electrochemical Cell Corrosion-Resistant Sealed and Non-Sealed
- Super Sealed Electrolytic Electrochemical Cell
- Electrolytic Electrochemical Cell with Five-Port
- H Type Electrolytic Cell Triple Electrochemical Cell
- Electrolytic Electrochemical Cell Gas Diffusion Liquid Flow Reaction Cell
Related Articles
- The Architecture of Certainty: Mastering Control in Multifunctional Electrolytic Cells
- Advanced Electrolytic Cell Techniques for Cutting-Edge Lab Research
- Electrolytic Batteries A Solution to the Growing Energy Demands
- The Art of the Non-Spontaneous: Precision in Electrolytic Circuits
- The Anchor of Truth: Why Physical Stability Defines Electrochemical Success




















