In the laboratory, we often obsess over the chemistry. We meticulously calculate molarities. We polish electrodes until they mirror our own exhaustion. We refine the code for the potentiostat.
But we frequently ignore the vessel itself.
This is a psychological blind spot. We view the electrolytic cell as merely a bucket—a passive container for the "real" science.
This assumption is dangerous. The cell is not a bucket; it is an interface. It is the boundary between the chaotic environment of the lab and the controlled universe of your experiment.
The success of that interface depends entirely on a few millimeters of glass. Specifically, the standard aperture specifications.
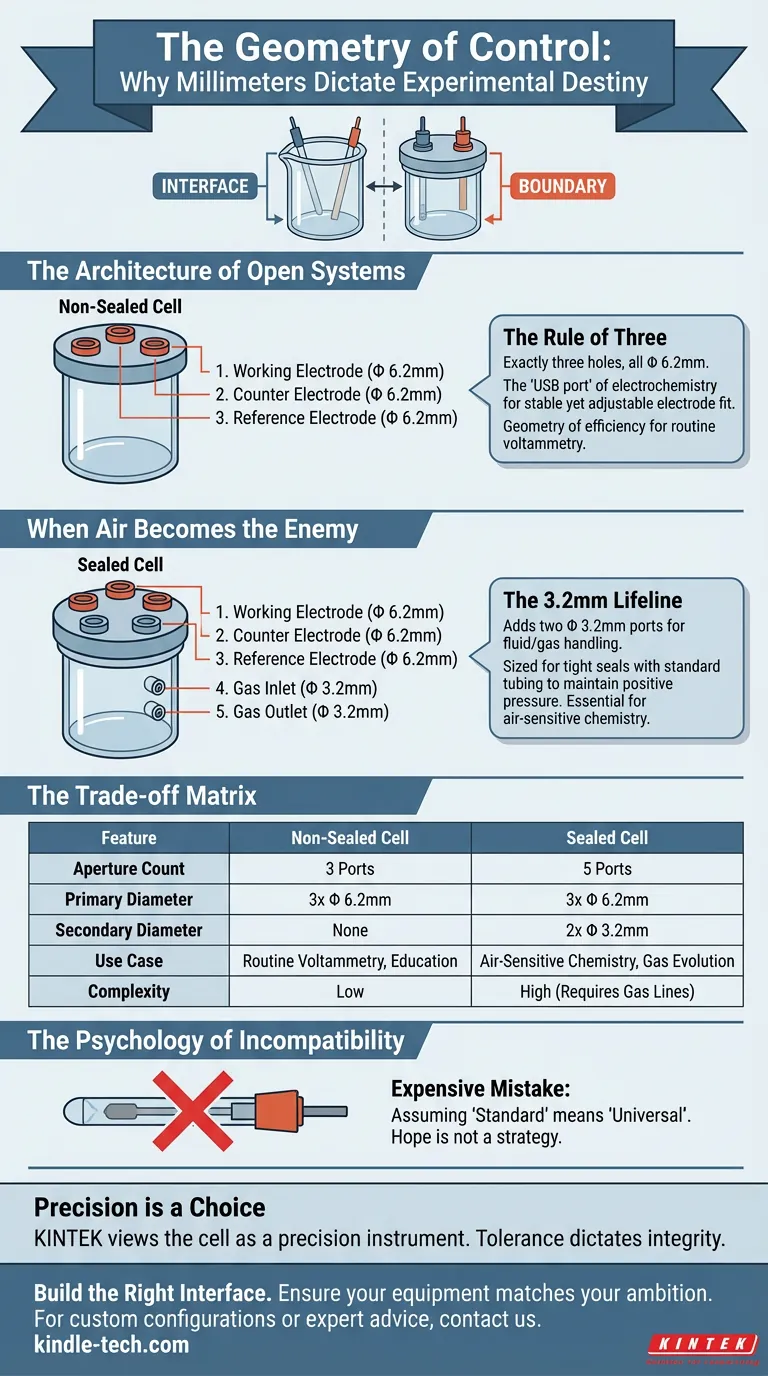
The Architecture of Open Systems
Most electrochemical experiments begin with the Non-Sealed Cell.
It represents the baseline of scientific inquiry. It is designed for robustness and accessibility. But its simplicity is deceptive. Its design relies on a rigid industry standard: The Φ 6.2mm aperture.
The Rule of Three
A standard non-sealed cell features exactly three holes, all drilled to a diameter of Φ 6.2mm.
This is not an arbitrary number. It is the "USB port" of electrochemistry.
- Working Electrode
- Counter Electrode
- Reference Electrode
The Φ 6.2mm dimension allows these standard electrodes to slide in with a fit that is secure enough to be stable, yet loose enough to allow for adjustment.
If you are running cyclic voltammetry on stable compounds, this is your workhorse. It is the geometry of efficiency.
When Air Becomes the Enemy
The moment your chemistry becomes sensitive to oxygen, or involves gaseous reactants, the "open bucket" philosophy fails.
You need an airlock. You need a Sealed Cell.
Here, the architecture changes. The geometry becomes more complex because the requirements for control have increased. The standard sealed cell retains the three Φ 6.2mm ports for the electrodes, but adds a critical new dimension: The Φ 3.2mm port.
The 3.2mm Lifeline
A sealed cell typically adds two holes of Φ 3.2mm.
While the large ports handle the solid interfaces (electrodes), these smaller ports handle the fluids (gases).
- Inlet: For sparging inert gases like Nitrogen or Argon to kill dissolved oxygen.
- Outlet: To vent the system without backflow.
Why 3.2mm? It is sized perfectly for standard PTFE or thin plastic tubing. It creates a tight seal necessary to maintain positive pressure. If these holes were 6.2mm, the tubing would flail, the seal would fail, and the atmosphere would intrude.
The Psychology of Incompatibility
The most expensive mistake in a lab is rarely an explosion. It is the "small" incompatibility that halts research for three weeks.
We see this often. A researcher buys a cell assuming "standard" means "universal." They arrive with a custom reference electrode that is 6.5mm wide.
It doesn’t fit.
Or they attempt to force a gas line into an electrode port, using Parafilm and hope to create a seal.
Hope is not a strategy.
The Trade-off Matrix
Understanding the anatomy of your cell allows you to make the right trade-off between control and complexity.
| Feature | Non-Sealed Cell | Sealed Cell |
|---|---|---|
| Aperture Count | 3 Ports | 5 Ports |
| Primary Diameter | 3x Φ 6.2mm | 3x Φ 6.2mm |
| Secondary Diameter | None | 2x Φ 3.2mm |
| Use Case | Routine Voltammetry, Education | Air-Sensitive Chemistry, Gas Evolution |
| Complexity | Low | High (Requires Gas Lines) |
Precision is a Choice
At KINTEK, we view the electrolytic cell as a precision instrument.
We understand that the tolerance of a glass port dictates the integrity of a sealed environment. We know that a Φ 6.2mm hole must actually be Φ 6.2mm, not Φ 6.0mm and not Φ 6.5mm.
If your research fits the industry standards, our off-the-shelf non-sealed and sealed cells provide immediate, reliable infrastructure for your work.
However, science often pushes the boundaries of "standard."
- Do you use oversized electrodes?
- Do you need a Luggin capillary for IR drop compensation?
- Do you need additional ports for temperature sensors?
If so, the standard geometry will fight you.
Build the Right Interface
Don't let a millimeter of glass dictate the limits of your discovery.
Whether you need the robust simplicity of a standard non-sealed cell or the rigorous atmosphere control of a sealed system, ensure your equipment matches your ambition.
If you are unsure if your current electrodes will fit, or if you need a custom configuration to accommodate a complex sensor array, reach out to us. We speak the language of precision.
Visual Guide
Related Products
- Electrolytic Electrochemical Cell with Five-Port
- H Type Electrolytic Cell Triple Electrochemical Cell
- Electrolytic Electrochemical Cell Gas Diffusion Liquid Flow Reaction Cell
- Electrode Fixture for Electrochemical Experiments
- Double Layer Five-Port Water Bath Electrolytic Electrochemical Cell
Related Articles
- The Architecture of Control: Decoding the Five-Port Electrolytic Cell
- The Fragility of Precision: Mastering the Integrity of Five-Port Electrolytic Cells
- Electrochemistry The Science Behind Electrochemical Cells
- The Fragile Vessel of Truth: A Maintenance Manifesto for Electrolytic Cells
- Electrolytic Batteries A Solution to the Growing Energy Demands



















