The Silent Partner in the Lab
In science, we obsess over the variables we can see. We meticulously calibrate the potentiostat. We polish the working electrode until it mirrors our own exhaustion. We purify the electrolyte to the parts-per-billion level.
But often, we ignore the room where the reaction happens.
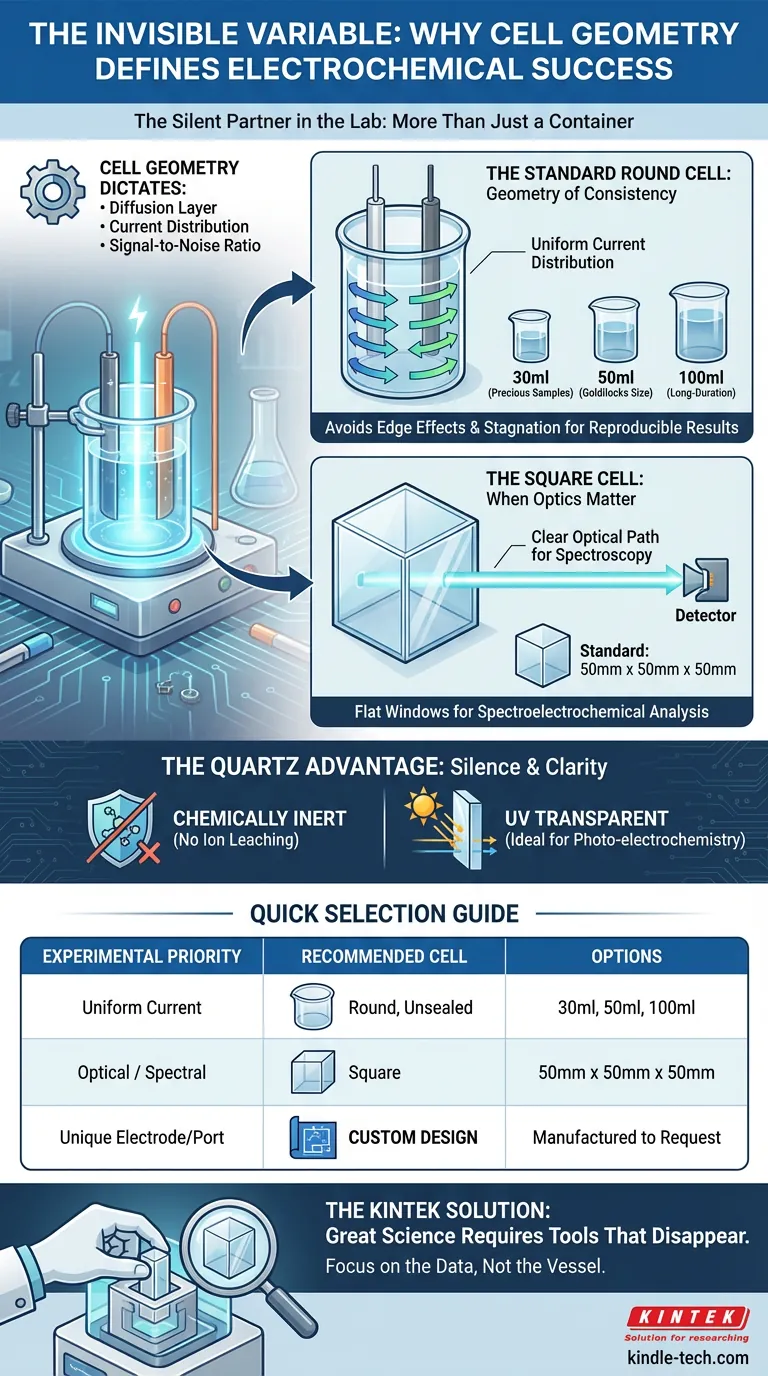
The electrolytic cell is frequently treated as a passive bucket—a mere container for the "real" science. This is a mistake. In electrochemistry, the vessel is a boundary condition. It defines the diffusion layer, dictates the current distribution, and controls the signal-to-noise ratio.
Choosing the right cell volume and dimension isn't just a purchasing decision; it is the first step in experimental design.
The Standard Round Cell: The Geometry of Consistency
For most general-purpose electrochemistry, the enemy is edge effects.
Sharp corners create areas of unequal current density. They cause stagnation points in the electrolyte. This is why the standard industry workhorse is the round, unsealed quartz cell.
The circular geometry promotes uniform current distribution between the working and counter electrodes. It is the path of least resistance for reproducible cyclic voltammetry.
Standard volumes are designed around the economy of reagents and electrode size:
- 30ml: Ideal for precious samples where electrolyte conservation is critical.
- 50ml: The "Goldilocks" size for standard setups, balancing volume with electrode space.
- 100ml: Necessary for larger electrodes or long-duration experiments where concentration changes must be minimized.
The Square Cell: When Optics Matter
Sometimes, the priority shifts from pure electrochemical kinetics to observation.
If you are running spectroelectrochemical experiments, a round cell becomes a nightmare of refraction and optical distortion. You cannot measure what you cannot clearly see.
To solve this, the standard shifts to a square quartz cell.
- Standard Dimension: 50mm x 50mm x 50mm.
This geometry provides the flat, parallel optical windows required for spectroscopic analysis. While it introduces corners (and thus potential current inhomogeneity), it offers the transparency required to correlate current flow with spectral changes.
The Quartz Advantage
Why are these standard cells constructed exclusively from quartz?
It comes down to two factors: silence and clarity.
Glass is a complex soup of oxides that can leach ions under extreme pH conditions, introducing noise to your data. Quartz is chemically inert. It remains silent, ensuring the current you measure comes from your analyte, not your container.
Furthermore, standard glass blocks UV light. Quartz allows it to pass. For any experiment involving photo-electrochemistry, quartz is not a luxury; it is a necessity.
The Trap of "One Size Fits All"
There is a psychological comfort in buying off the shelf. It feels safe. It feels standard.
But research operates at the edge of the known. Often, a standard 50ml cell is too large for a synthesized catalyst, or a 50mm square cell doesn't fit a specialized spectrometer.
Forcing an experiment into the wrong vessel introduces error. A cell that is too large wastes expensive reagents. A cell that is too small crowds electrodes, causing short circuits or turbulent flow.
This is where customization becomes the most rigorous scientific choice.
Most suppliers, including KINTEK, can manufacture cells based on specific drawings. If your protocol requires a unique path length, specific port placements, or non-standard volumes, the hardware should adapt to the science, not the other way around.
Quick Selection Guide
To simplify the decision process, match your primary experimental constraint to the cell type:
| Experimental Priority | Recommended Cell Type | Standard Options |
|---|---|---|
| Uniform Current Distribution | Round, Unsealed | 30ml, 50ml, 100ml |
| Optical/Spectral Analysis | Square | 50mm x 50mm x 50mm |
| Unique Electrode/Port Needs | Custom Design | Manufactured to Request |
The Solution
Great science requires tools that disappear.
When your equipment fits perfectly, you stop thinking about it. You stop worrying about leaks, optical distortion, or wasted reagent. You focus entirely on the data.
At KINTEK, we provide the foundational equipment that allows that focus. Whether you need the reliability of our standard quartz cells or a bespoke design for a novel breakthrough, we ensure the vessel matches the vision.
Contact Our Experts to discuss whether a standard volume or a custom dimension is right for your next experiment.
Visual Guide
Related Products
- Quartz Electrolytic Electrochemical Cell for Electrochemical Experiments
- Electrolytic Electrochemical Cell with Five-Port
- PTFE Electrolytic Cell Electrochemical Cell Corrosion-Resistant Sealed and Non-Sealed
- Electrolytic Electrochemical Cell for Coating Evaluation
- Flat Corrosion Electrolytic Electrochemical Cell
Related Articles
- The Architecture of Transparency: Mastering Safety and Precision in Quartz Electrolysis
- Electrochemical Cells: Generating Electricity and Driving Reactions
- The Geometry of Control: Why Millimeters Matter in Electrochemistry
- The Architecture of Silence: Why Quartz Defines Electrochemical Precision
- Benefits of Electrochemical Cells for Energy Storage




















