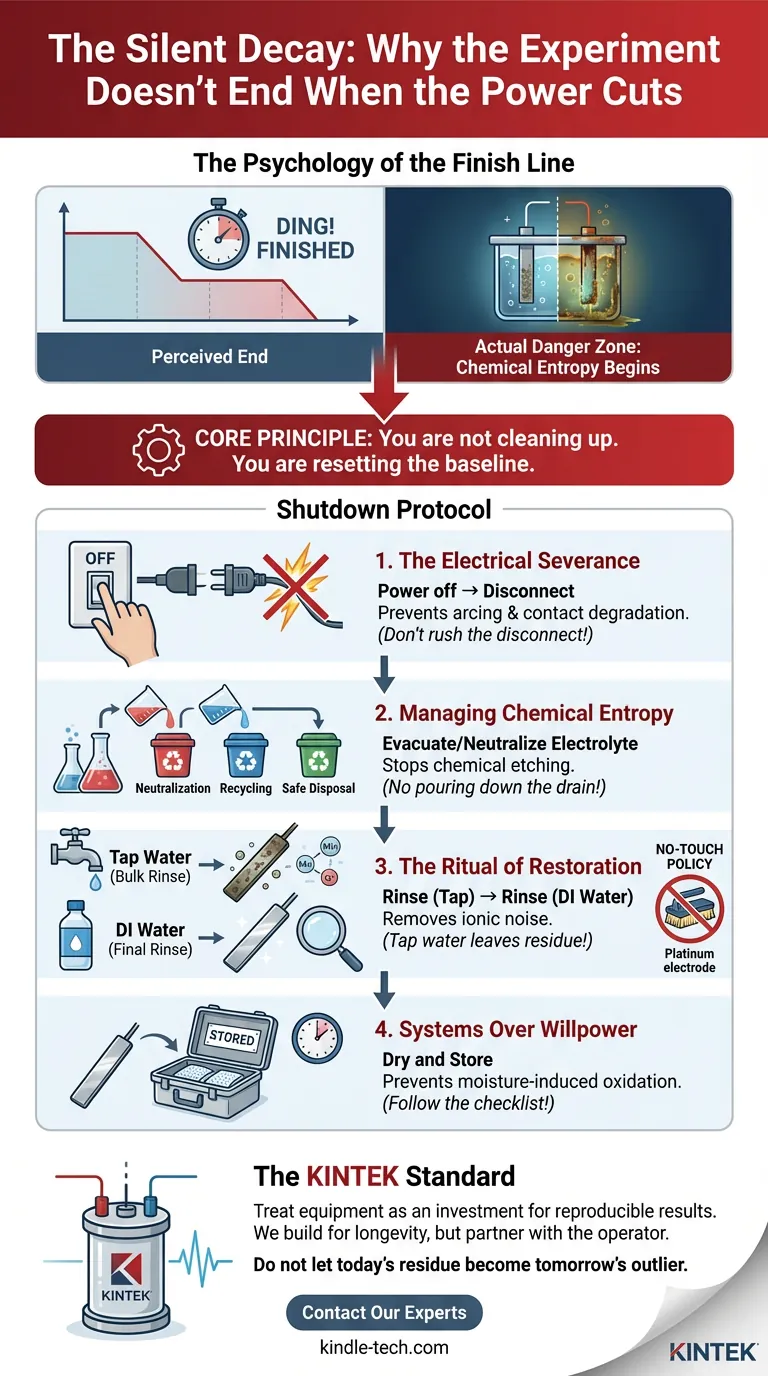
The Psychology of the Finish Line
There is a distinct psychological shift that happens the moment data collection stops.
The graph flattens. The timer dings. The reaction stabilizes. In the mind of the researcher, the work is finished. The temptation to rush—to strip the equipment, scribble the notes, and leave the lab—is overwhelming.
But in electrochemistry, this is the most dangerous moment.
It is dangerous not just for the operator, but for the data that will be collected next week. The integrity of an electrolytic cell is not defined by how it starts, but by how it ends.
The core principle of post-electrolysis procedures is simple: You are not cleaning up. You are resetting the baseline.
We must treat the shutdown sequence not as a chore, but as a systematic preservation of the future. Here is the engineering logic behind the shutdown.
The Electrical Severance
Electricity has momentum. Even when the intent to stop is there, the potential remains.
The most common error in laboratory shutdowns is disconnecting the circuit before killing the source. It feels efficient. It is actually a hazard.
The Rule: Turn off the power supply completely. Then, and only then, touch the connections.
If you disconnect a live circuit, you risk an electrical arc. This is not just a safety hazard for the operator; it is a trauma for the equipment. Arcing scars the connection points. It alters the resistance of the contacts.
Over time, these micro-scars accumulate. Your voltage readings become erratic. You blame the solution, but the fault lies in a sloppy shutdown months ago.
Managing Chemical Entropy
Once the power is cut, the cell is no longer a reactor. It is a storage vessel for decaying chemistry.
The electrolyte does not pause just because you turned off the switch. Depending on its composition, it may begin to crystallize, corrode the vessel walls, or react with atmospheric carbon dioxide.
Safe handling requires immediate decision-making based on chemical properties:
- Neutralization: For acidic or basic solutions.
- Recycling: For valuable metallic ions.
- Disposal: Strictly following environmental protocols.
Pouring reactive chemicals down the drain is not just an environmental crime; it is a failure of laboratory discipline that creates unpredictable variables in the plumbing infrastructure of your facility.
The Ritual of Restoration
Corrosion is the enemy of reproducibility.
If you leave an electrolytic cell unwashed for an hour, you allow residue to solidify. If you leave it overnight, you invite surface degradation.
The goal of cleaning is to erase the history of the previous run.
The Water Hierarchy
You cannot simply rinse and leave. The cleaning process follows a hierarchy of purity:
- Tap Water: Removes the bulk.
- Distilled/Deionized Water: The crucial second step.
Tap water contains ions—calcium, magnesium, chloride. If you let tap water dry on your electrodes, you are depositing impurities. You are essentially creating a new, unintentional alloy on the surface of your sensor.
Always finish with deionized water.
The "No-Touch" Policy
Electrodes are precision instruments, not dirty dishes.
Never use abrasive tools. A metal brush on a platinum electrode is catastrophic. It creates micro-scratches that increase surface area unpredictably, altering current density in future experiments.
If the residue is stubborn, use chemistry (dilute acids or bases), not physics (scrubbing).
Systems Over Willpower
Atul Gawande, in The Checklist Manifesto, argues that errors occur not because of ignorance, but because of ineptitude—failing to apply what we know.
In the lab, fatigue is the driver of ineptitude. By the end of the day, willpower is low.
To combat this, we rely on a rigid shutdown protocol. It removes the need for decision-making when you are tired.
| Sequence | The Action | The Engineering "Why" |
|---|---|---|
| 1. Sever | Power off → Disconnect | Prevents arcing and contact degradation. |
| 2. Evacuate | Remove/Neutralize Electrolyte | Stops chemical etching of the cell body. |
| 3. Reset | Rinse (Tap) → Rinse (DI Water) | Removes ionic noise from future data. |
| 4. Preserve | Dry and Store | Prevents moisture-induced oxidation during downtime. |
The KINTEK Standard
The difference between a lab that produces consistent results and one that struggles with "phantom errors" often comes down to equipment care.
But it also starts with the equipment itself.
At KINTEK, we design our electrolytic cells and lab consumables with the understanding that the lab environment is harsh. We build for longevity, but even the best engineering requires a partnership with the operator.
When you treat your equipment as an investment rather than a disposable tool, you secure the accuracy of your next discovery.
Do not let today's residue become tomorrow's outlier.
Our experts can help you select equipment designed for durability and ease of maintenance, ensuring your workflow remains seamless. Contact Our Experts to discuss how to upgrade your laboratory setup today.
Visual Guide
Related Products
- PTFE Electrolytic Cell Electrochemical Cell Corrosion-Resistant Sealed and Non-Sealed
- Super Sealed Electrolytic Electrochemical Cell
- Electrolytic Electrochemical Cell with Five-Port
- H Type Electrolytic Cell Triple Electrochemical Cell
- Electrolytic Electrochemical Cell Gas Diffusion Liquid Flow Reaction Cell
Related Articles
- The Art of the Non-Spontaneous: Precision in Electrolytic Circuits
- Advanced Electrolytic Cell Techniques for Cutting-Edge Lab Research
- The Art of Resistance: Why Your Electrolytic Cell Needs Breathing Room
- Electrolytic Batteries A Solution to the Growing Energy Demands
- Electrolytic Cell Design and Standards




















