The Illusion of the Knob
There is a temptation in laboratory work to view control as a simple mechanical act. You turn a dial, the needle moves, and the machine obeys.
In electrochemistry, this view is dangerous.
An electrolytic cell is not merely a machine; it is a chaotic environment where you are attempting to force nature to move backward. You are using energy to drive non-spontaneous reactions. To do this successfully, you cannot simply "turn it on."
You must act as an architect of the atomic environment.
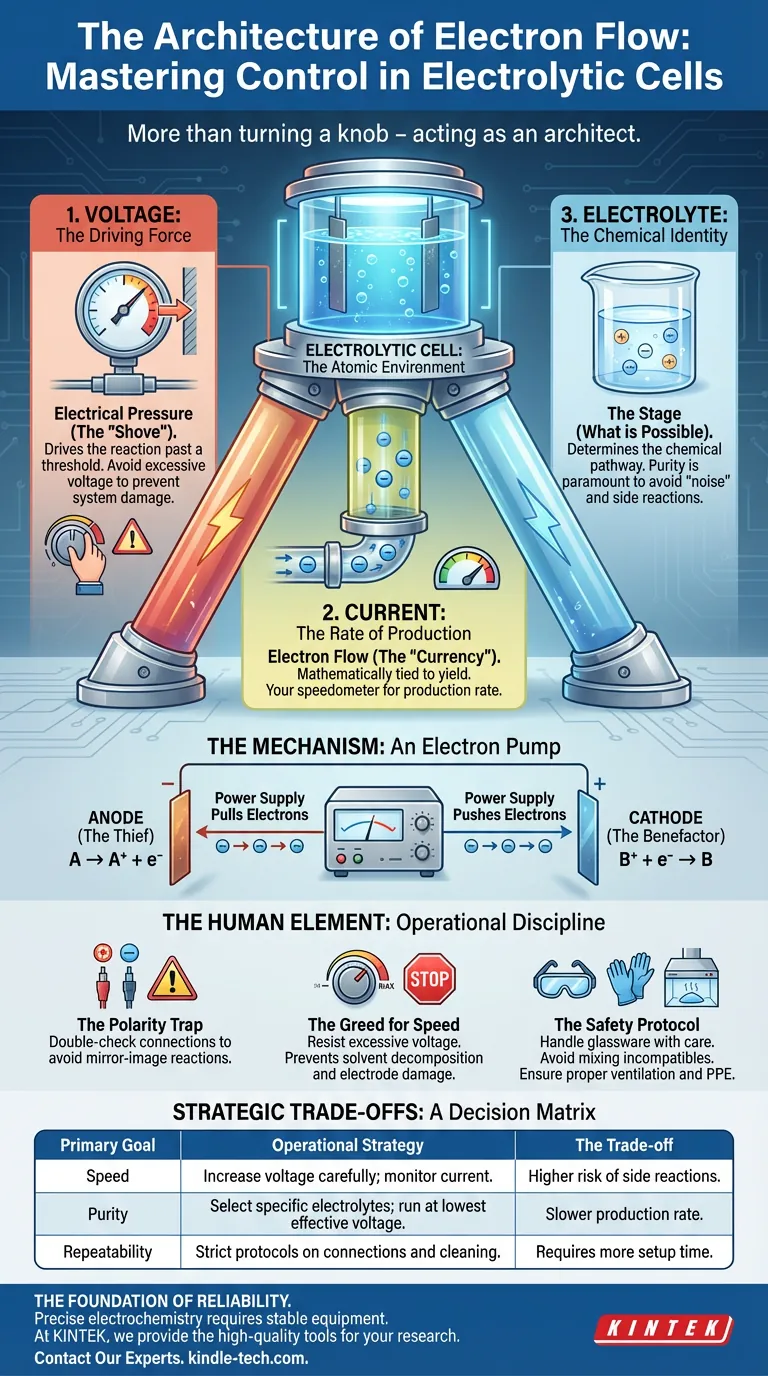
Control in this context is a tripod. It rests on three non-negotiable pillars: Voltage, Current, and Composition. If you ignore one, the structure collapses.
The Three Levers of Influence
To master the output of your cell, you must understand the distinct role of each variable. They are interconnected, but they are not the same.
1. Voltage: The Driving Force
Think of voltage as electrical pressure. It is the shove required to get the reaction moving.
Without sufficient voltage, nothing happens. The barrier to the reaction remains too high. However, more is not always better. Increasing voltage increases the rate, but only up to a threshold. Past that point, you are no longer driving the reaction—you are damaging the system.
2. Current: The Rate of Production
Current is the flow of electrons. In the economy of the electrolytic cell, electrons are the currency.
The amount of product you create is mathematically tied to the current. If you want a higher yield, you need more charge passing through the system over time. Current is your speedometer.
3. Electrolyte: The Chemical Identity
The electrolyte is the stage where the play happens. It determines what reaction is chemically possible.
If voltage and current are the engine, the electrolyte is the destination. Purity here is paramount. A contaminated electrolyte introduces "noise" into the system, leading to side reactions that ruin the final product.
The Mechanism: An Electron Pump
To control the cell, you must visualize what is happening at the electrodes. The external power supply acts as a pump, creating an imbalance that nature desperately wants to correct.
- At the Anode (The Thief): The power supply pulls electrons away. This forces the electrolyte species to lose electrons (oxidation).
- At the Cathode (The Benefactor): The power supply pushes electrons in. This forces the species to gain electrons (reduction).
Your job is to regulate this pump so that the exchange happens exactly as planned, without overwhelming the chemical capacity of the solution.
The Human Element: Operational Discipline
The biggest variable in any experiment is the operator. Theoretical knowledge fails when met with practical negligence.
In surgery, a checklist saves lives. In the lab, a protocol saves experiments.
The Polarity Trap
Reversing the anode and cathode is a simple error with profound consequences. It creates a mirror-image reaction, producing the wrong substances at the wrong poles. Check your connections twice.
The Greed for Speed
There is a psychological urge to crank the voltage to finish faster. Resist it.
Excessive voltage often leads to the decomposition of the solvent (usually water) or the degradation of the electrode surface. You trade purity for speed, and usually, you lose both.
The Safety Protocol
Chemistry is unforgiving of complacency.
- Glassware: Handle the cell with care. Scratches from metal brushes weaken the structural integrity.
- Mixtures: Never mix acids and bases during cleaning. The resulting exothermic reaction can shatter the vessel.
- Protection: Corrosive electrolytes and toxic gases demand proper ventilation (fume hoods) and PPE.
Strategic Trade-offs
Perfection is impossible. Optimization is the goal. Your control strategy should shift based on your specific objective.
Use this decision matrix to guide your setup:
| Primary Goal | Operational Strategy | The Trade-off |
|---|---|---|
| Speed | Increase voltage carefully; monitor current. | Higher risk of side reactions. |
| Purity | Select specific electrolytes; run at lowest effective voltage. | Slower production rate. |
| Repeatability | Strict protocols on connections and cleaning. | Requires more setup time. |
The Foundation of Reliability
You cannot control a reaction if you cannot trust your tools.
Precise electrochemistry requires equipment that provides stable voltage, consistent current, and durable materials that withstand corrosive environments without contaminating your results.
At KINTEK, we understand the engineer's romance with precision. We specialize in providing the high-quality lab equipment and consumables that serve as the silent partner in your research. When the hardware is reliable, you are free to focus on the science.
Visual Guide
Related Products
- PTFE Electrolytic Cell Electrochemical Cell Corrosion-Resistant Sealed and Non-Sealed
- Super Sealed Electrolytic Electrochemical Cell
- Electrolytic Electrochemical Cell with Five-Port
- H Type Electrolytic Cell Triple Electrochemical Cell
- Electrolytic Electrochemical Cell Gas Diffusion Liquid Flow Reaction Cell
Related Articles
- The Silent Geometry of Voltage: Respecting the Limits of Electrolysis
- The Art of the Non-Spontaneous: Precision in Electrolytic Circuits
- Advanced Electrolytic Cell Techniques for Cutting-Edge Lab Research
- The Architecture of Certainty: Mastering Control in Multifunctional Electrolytic Cells
- Electrolytic Cell Design and Standards




















