The Invisible variable
In any experiment, there is the variable you are testing, and there are the variables that test you.
We often obsess over the electrode or the electrolyte. We calculate currents and potentials down to the microvolt. But we rarely pause to consider the vessel holding it all together.
This is a blind spot.
If your containment vessel interacts with your solution, your data is compromised. The background noise becomes the signal. To get the truth, the environment must be "silent."
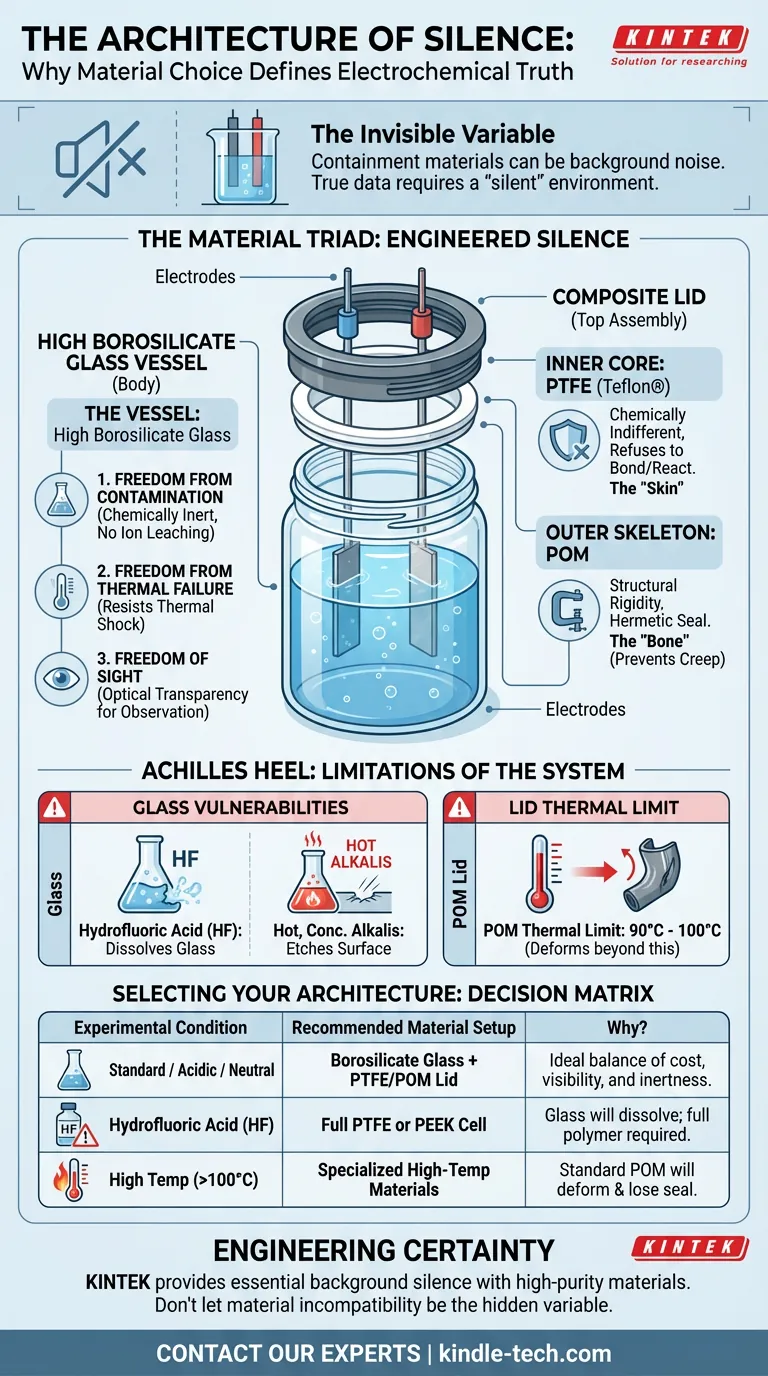
In coating evaluation electrolytic cells, this silence is engineered through a specific triad of materials: High Borosilicate Glass, PTFE, and POM.
It is not a random selection. It is a deliberate architectural choice designed to separate the observer from the reaction.
The Vessel: High Borosilicate Glass
The body of the cell is the stage. It needs to be there, but it also needs to disappear.
We use High Borosilicate Glass for the cell body. Standard glass wouldn't survive the rigors of the lab; plastic might obscure the view. Borosilicate offers three distinct "freedoms" to the engineer:
- Freedom from Contamination: It is chemically inert to most acids and organic solvents. It does not leach ions. It ensures that the corrosion you measure is from your sample, not your jar.
- Freedom from Thermal Failure: It resists thermal shock. You can push temperature boundaries without the catastrophic cracking associated with soda-lime glass.
- Freedom of Sight: Data isn't just digital. Optical transparency allows you to see the bubbles, the color shifts, and the delamination.
The Lid: A Study in Duality
The lid is where the engineering gets romantic. It has to solve a paradox: it must be chemically invincible on the inside, but mechanically rigid on the outside.
One material cannot do both well. So, we use a composite approach.
1. The Inner Core: PTFE (Teflon®)
The surface facing the chemical storm is Polytetrafluoroethylene.
PTFE is the ultimate introvert of the chemical world. It refuses to bond or react with almost anything. By lining the inner core with PTFE, we ensure that the aggressive vapors and splashing electrolytes meet a wall of indifference.
2. The Outer Skeleton: POM (Polyoxymethylene)
PTFE has a flaw: it is soft. It "creeps" under pressure. You cannot thread a screw tightly into it and expect it to hold forever.
Enter POM.
Used for the external screw caps and structural rings, POM is the "bone" to the PTFE "skin." It provides the stiffness required to clamp down on the electrodes and maintain a hermetic seal against the atmosphere.
The "Achilles Heel" of Glass
Despite this careful engineering, no system is perfect. Understanding the limits of your equipment is just as important as understanding its capabilities.
The "silence" of borosilicate glass is broken by two specific conditions:
- Hydrofluoric Acid (HF): This acid eats glass. Using it in a standard cell will destroy your equipment and ruin your data.
- Hot, Concentrated Alkalis: Strong bases at high temperatures will etch the glass surface over time.
Furthermore, while the glass can handle high heat, the lid cannot. The POM components typically have a thermal limit around 90°C to 100°C. Pushing beyond this transforms a precision instrument into a warped piece of plastic.
Selecting Your Architecture
The material of your cell dictates the validity of your results.
If you choose the wrong vessel, you aren't measuring the coating; you are measuring the breakdown of your equipment.
Here is the decision matrix for the pragmatic researcher:
| Experimental Condition | Recommended Material Setup | Why? |
|---|---|---|
| Standard / Acidic / Neutral | Borosilicate Glass + PTFE/POM Lid | Ideal balance of cost, visibility, and inertness. |
| Hydrofluoric Acid (HF) | Full PTFE or PEEK Cell | Glass will dissolve; full polymer construction is required. |
| High Temp (>100°C) | Specialized High-Temp Materials | Standard POM fittings will deform and lose seal integrity. |
Engineering Certainty
At KINTEK, we believe that the best lab equipment is the kind you don't have to worry about.
Our electrolytic cells are crafted to provide that essential background silence. We use high-purity borosilicate glass and precision-machined PTFE to ensure that when you look at your data, you are seeing the chemistry, not the container.
Do not let material incompatibility become the hidden variable in your research.
Let us help you select the perfect cell architecture for your specific application. Contact Our Experts
Visual Guide
Related Products
- CF Ultra-High Vacuum Observation Window Window Flange High Borosilicate Glass Sight Glass
- KF Ultra-High Vacuum Observation Window 304 Stainless Steel Flange High Borosilicate Glass Sight Glass
- Optical Window Glass Substrate Wafer Single Double Sided Coated K9 Quartz Sheet
- High Temperature Resistant Optical Quartz Glass Sheet
- KF Ultra-High Vacuum Observation Window Stainless Steel Flange Sapphire Glass Sight Glass
Related Articles
- X-Ray Fluorescence Spectrometer: Powder Compression Method for Sample Preparation
- Common Instrumentation and Operations in Organic Synthesis
- Comprehensive Guide to Optical Windows: Material Properties and Surface Specifications
- The Architecture of Invisible Containment: Why Material Choice Defines Electrochemical Precision
- The Rise of Glass Substrates in Advanced Semiconductor Packaging









