The Illusion of Simplicity
In the laboratory, we often fixate on the complexity of the electrolyte. We measure concentrations to the microgram. We obsess over temperature gradients. We treat the chemical solution like a delicate recipe that requires a master chef’s touch.
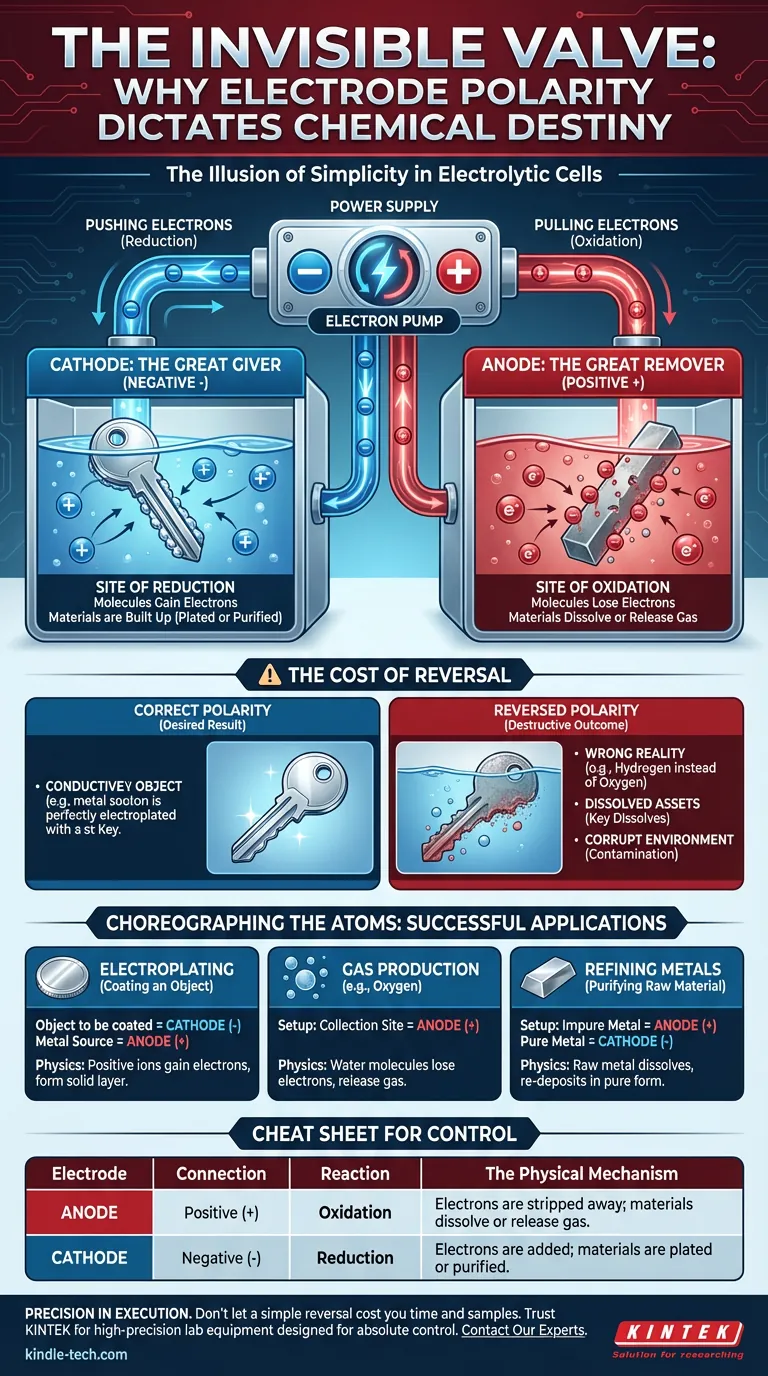
Yet, we frequently overlook the "plumbing" of the experiment: the wires.
Connecting an electrolytic cell seems trivial. Red wire here, black wire there. It feels like plugging in a toaster—does it really matter which way it goes?
In electrolysis, it matters more than anything else.
Electrode polarity is not merely a connection detail. It is the command and control center of your experiment. It is the difference between creating a masterpiece and dissolving your equipment.
The Electron Pump
To understand polarity, you have to stop thinking of the power supply as a battery and start thinking of it as a pump.
Unlike a galvanic cell, where chemistry creates electricity, an electrolytic cell uses electricity to force chemistry to happen against its will. The reaction is non-spontaneous. It needs a shove.
The power supply provides that shove, but strictly in one direction:
- It pulls electrons from one side.
- It pushes electrons to the other.
This flow dictates the identity of your electrodes. The labels "Anode" and "Cathode" are not geographic locations in your beaker; they are functional roles defined entirely by this flow.
The Anode: The Great Remover
Connected to the Positive (+) terminal, the anode is where the power supply rips electrons away from chemical species.
This is the site of Oxidation. Molecules near this surface lose electrons. They are stripped down.
The Cathode: The Great Giver
Connected to the Negative (-) terminal, the cathode is where the power supply forces an excess of electrons onto the surface.
This is the site of Reduction. Molecules near this surface gain electrons. They are built up.
The Cost of Reversal
What happens when you swap the wires?
In many areas of life, a reversed connection just means the device doesn't turn on. In electrolysis, the consequences are far more active—and often destructive.
When you reverse polarity, you don't pause the reaction. You invert the universe of your beaker.
1. You manufacture the wrong reality
If you intended to generate oxygen but swapped the terminals, you might generate hydrogen instead. The chemical logic is perfectly inverted. You aren't getting "no result"; you are getting the "anti-result."
2. You dissolve your assets
Imagine you are electroplating a vintage key with copper. You want the copper ions to plate onto the key.
If you reverse the polarity, the key becomes the anode (the site of oxidation). instead of receiving a shiny coat of copper, the key itself begins to dissolve into the solution. You aren't preserving the object; you are actively destroying it.
3. You corrupt the environment
Forcing oxidation on an electrode not designed for it leads to rapid corrosion. This doesn't just ruin the electrode; it floods your carefully prepared electrolyte with contaminants, invalidating the entire experiment.
Choreographing the Atoms
Successful electrolysis requires you to be an architect of atomic movement. You must decide where the electrons go before you flip the switch.
Use this logic to assign your polarity:
-
The Goal: Electroplating
- The Setup: The object to be coated must be the Cathode (-).
- The Physics: You need positive metal ions to gain electrons and turn into solid metal on the object's surface.
-
The Goal: Gas Production (e.g., Oxygen)
- The Setup: The collection site must be the Anode (+).
- The Physics: Water molecules must lose electrons (oxidize) to release oxygen gas.
-
The Goal: Refining Metals
- The Setup: The impure metal must be the Anode (+).
- The Physics: The raw metal must dissolve (oxidize) into the solution so it can be re-deposited in pure form elsewhere.
The Summary of Control
Here is the cheat sheet for controlling your chemical destiny:
| Electrode | Connection | Reaction | The Physical Mechanism |
|---|---|---|---|
| Anode | Positive (+) | Oxidation | Electrons are stripped away; materials dissolve or release gas. |
| Cathode | Negative (-) | Reduction | Electrons are added; materials are plated or purified. |
Precision in Execution
There is a certain romance in electrolysis. You are reaching into the molecular world and forcing atoms to march in the direction you choose.
But that control relies on the integrity of your setup. Even the best theoretical understanding fails if the equipment is unreliable or the connections are poor.
At KINTEK, we understand that the line between a breakthrough and a failed experiment is often as thin as a wire connection. We specialize in high-precision lab equipment and consumables designed to give you absolute control over your electrochemical processes.
Whether you are refining precious metals or conducting sensitive research, our tools ensure that when you command the electrons to flow, they go exactly where they are supposed to.
Don't let a simple reversal cost you time and samples. Contact Our Experts
Visual Guide
Related Products
- PTFE Electrolytic Cell Electrochemical Cell Corrosion-Resistant Sealed and Non-Sealed
- Super Sealed Electrolytic Electrochemical Cell
- Electrolytic Electrochemical Cell with Five-Port
- H Type Electrolytic Cell Triple Electrochemical Cell
- Electrolytic Electrochemical Cell Gas Diffusion Liquid Flow Reaction Cell
Related Articles
- The Architecture of Certainty: Mastering Control in Multifunctional Electrolytic Cells
- Electrolytic Batteries A Solution to the Growing Energy Demands
- The Art of Resistance: Why Your Electrolytic Cell Needs Breathing Room
- The Anchor of Truth: Why Physical Stability Defines Electrochemical Success
- The Silent Partner: Why Material Choice in Electrochemistry is a Matter of Trust




















