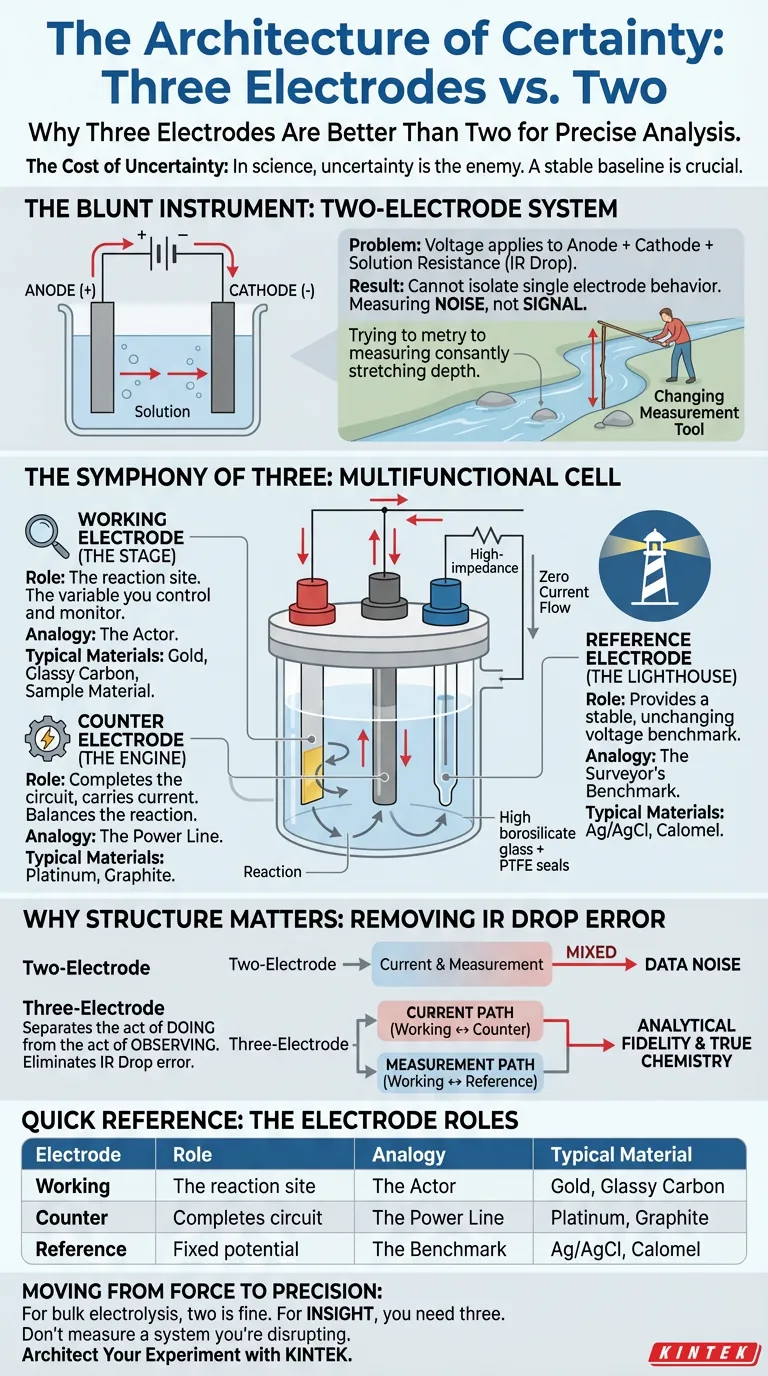
The Cost of Uncertainty
In science, as in life, uncertainty is the enemy.
We crave a stable baseline. We need to know that a change in outcome is caused by a change in variable, not a flaw in the measurement tool itself.
In the world of electrochemistry, this distinction is often blurred.
Imagine trying to measure the depth of a river while the stick you are using is constantly changing length based on the speed of the current. That is the fundamental flaw of a simple, two-electrode system when used for precise analysis.
To solve this, engineers and chemists developed a more elegant architecture: the three-electrode multifunctional electrolytic cell. It is a system designed not just to make reactions happen, but to understand exactly how they happen.
The Blunt Instrument: Two-Electrode Systems
To understand the solution, we must first appreciate the problem.
A standard electrolytic cell has two actors:
- The Anode
- The Cathode
You apply voltage across them to drive a reaction. This works perfectly for bulk electrolysis—like plating gold or producing chlorine. You pump in energy, and you get a product.
But if your goal is analysis—measuring reaction kinetics or potential thresholds—this system fails.
Why? Because the voltage you apply is consumed by three things simultaneously: the reaction at the anode, the reaction at the cathode, and the resistance of the solution itself (the IR drop).
You cannot isolate the behavior of a single electrode. You are measuring the noise of the entire system, not the signal of your specific reaction.
The Symphony of Three
The "multifunctional" cell introduces a third player to solve the problem of noise.
It separates the act of doing (driving current) from the act of observing (measuring potential). It transforms a blunt instrument into a scalpel.
Here is how the three roles interact.
1. The Working Electrode (The Stage)
This is where the story happens.
Whether you are studying corrosion, testing a catalyst, or developing a sensor, the Working Electrode is the site of that specific reaction.
It is the variable you want to control. In a KINTEK cell, this is the component you monitor with obsession. Everything else in the cell exists to ensure this electrode can tell its truth without interference.
2. The Counter Electrode (The Engine)
Electricity requires a circuit. Electrons leaving the working electrode must go somewhere.
The Counter Electrode (or auxiliary electrode) acts as this sink or source. It balances the reaction.
It is the "mule" of the system. It carries the current so the measurement tools don’t have to. Usually made of inert platinum or graphite, it is designed to be invisible to the chemistry but essential to the physics.
3. The Reference Electrode (The Lighthouse)
This is the most critical component for precision.
The Reference Electrode has one job: to provide a stable, unchanging voltage benchmark (like Ag/AgCl).
It connects to the system through a high-impedance path. This means almost zero current flows through it. Because no current flows, its potential never changes, regardless of what chaos is happening in the rest of the cell.
It acts as a fixed point in a shifting sea. It allows the potentiostat to measure the difference between the Working Electrode and this fixed point, ignoring the voltage drops elsewhere in the solution.
Why Structure Matters
The elegance of the three-electrode system is that it removes the "IR drop" error from your data.
By separating the current path (Working ↔ Counter) from the measurement path (Working ↔ Reference), you achieve analytical fidelity.
However, the quality of the data is only as good as the physical integrity of the cell.
Material Integrity
In high-precision labs, contamination is a subtle disaster. This is why multifunctional cells are crafted from:
- High Borosilicate Glass: For optical clarity and chemical resistance.
- PTFE (Teflon) Seals: To prevent polymer leaching or corrosion.
The cell body must be an inert vessel. It should contain the experiment, not participate in it.
Quick Reference: The Electrode Roles
| Electrode | Role | Analogy | Typical Material |
|---|---|---|---|
| Working | The reaction site | The Actor on Stage | Gold, Glassy Carbon, Sample Material |
| Counter | Completes the circuit | The Power Line | Platinum, Graphite |
| Reference | Provides fixed potential | The Surveyor's Benchmark | Ag/AgCl, Calomel |
Moving From Force to Precision
If your goal is simply to make a gas or plate a metal, a two-electrode system is fine. It is cost-effective and robust.
But if your goal is insight—if you need to know the exact potential where a molecule oxidizes, or the precise rate of a corrosion mechanism—you cannot rely on a two-electrode setup.
The three-electrode system is the standard for modern research because it acknowledges a fundamental truth: you cannot accurately measure a system that you are simultaneously disrupting.
By isolating the reference point, you gain the ability to see the chemistry as it truly is.
Architect Your Experiment
At KINTEK, we believe that the equipment you use should be as precise as the questions you ask. Whether you need the inert stability of high-quality glass or the exactitude of a calibrated reference electrode, our systems are built to eliminate the noise so you can focus on the signal.
Visual Guide
Related Products
- H Type Electrolytic Cell Triple Electrochemical Cell
- Three-dimensional electromagnetic sieving instrument
- Gold Disc Electrode
- Rotating Platinum Disk Electrode for Electrochemical Applications
- Reference Electrode Calomel Silver Chloride Mercury Sulfate for Laboratory Use
Related Articles
- The Art of Resistance: Why Your Electrolytic Cell Needs Breathing Room
- The Silent Geometry of Voltage: Respecting the Limits of Electrolysis
- The Silent Partner: Why Material Choice in Electrochemistry is a Matter of Trust
- The Invisible Variable: Mastering the Art of Electrolytic Cell Integrity
- Applications of H-Type Electrolytic Cell in Metal Extraction











