Chemistry is often taught as a series of equations. A + B yields C.
But in the laboratory, chemistry is actually a battle against variables.
A one-degree fluctuation in temperature can alter a reaction rate. A trace amount of oxygen can poison a reduction surface. The "perfect" experiment on paper falls apart because the physical environment wasn't compliant.
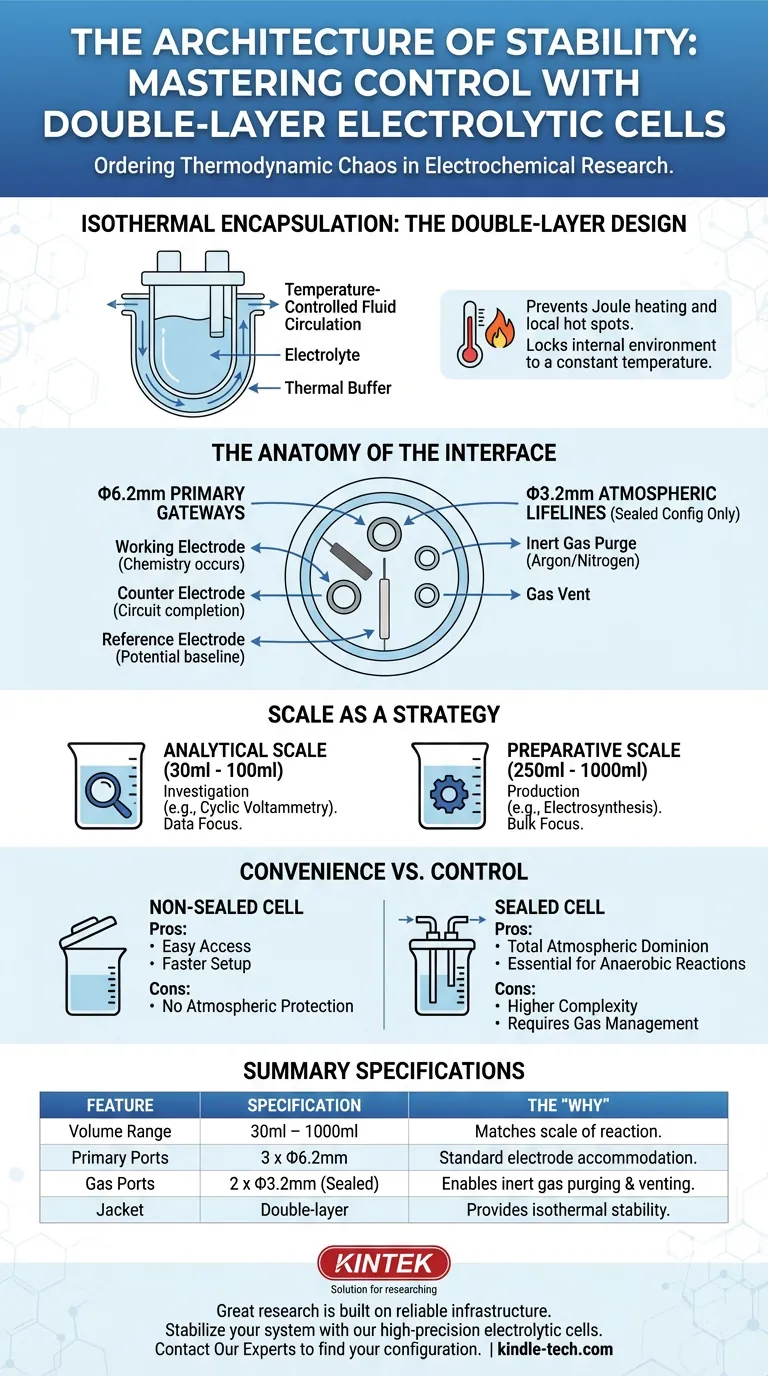
This is why the Double-Layer Water-Bath Electrolytic Cell is more than just glassware. It is an engineered environment designed to impose order on thermodynamic chaos.
The Illusion of Ambient Control
We tend to assume the room temperature is constant. It isn't.
More importantly, electrochemical reactions are rarely thermally neutral. They generate heat—specifically Joule heating—as current passes through the electrolyte. Without intervention, your electrolyte warms up, the viscosity changes, diffusion rates shift, and your data becomes a moving target.
The double-layer design solves this through isothermal encapsulation.
By circulating temperature-controlled fluid through an outer jacket, the cell acts as a massive thermal buffer. It prevents local hot spots on electrode surfaces and locks the internal environment to a specific temperature. It turns a variable into a constant.
The Anatomy of the Interface
A cell is defined by how it interacts with the outside world. This interface is governed by its aperture configuration.
The standard industry design is not random; it is a calculated layout intended for the classic Three-Electrode System.
The Primary Gateways (Φ6.2mm)
Whether sealed or non-sealed, the cell typically features three primary ports with a 6.2mm diameter. These are the docking stations for your core sensors:
- The Working Electrode: Where the chemistry happens.
- The Counter Electrode: Completing the circuit.
- The Reference Electrode: Providing the potential baseline (often via a Luggin capillary).
The Atmospheric Lifelines (Φ3.2mm)
This is where the distinction between "good enough" and "precision" lies.
In a sealed configuration, the cell includes two additional, smaller ports with a 3.2mm diameter.
These are not vestigial. They are the lifelines for gas management. They allow you to purge the system with inert gases like argon or nitrogen, removing oxygen for anaerobic experiments. They allow the cell to breathe without becoming contaminated.
Scale as a Strategy
The volume of your cell is not just about how much liquid you have. It is a strategic decision about the intent of your research.
Standard volumes range from 30ml to 1000ml, but the utility shifts across this spectrum:
-
Analytical Scale (30ml - 100ml): These are built for investigation. You use them for cyclic voltammetry or when your electrolyte is expensive (e.g., ionic liquids). The goal is data, not product.
-
Preparative Scale (250ml - 1000ml): These are built for production. When moving to bulk electrolysis or electrosynthesis, you need a larger thermal mass and reservoir to prevent rapid depletion of reactants.
The Trade-off: Convenience vs. Control
Every engineer knows that optimization requires trade-offs. The same applies to selecting your cell type.
The Non-Sealed Cell
- Pros: Easy access, faster setup, robust for air-stable reactions.
- Cons: No protection against atmospheric interference.
The Sealed Cell
- Pros: Total atmospheric dominion. Essential for reduction reactions sensitive to oxygen.
- Cons: Higher complexity. Requires gas lines, septa, and disciplined handling.
Summary Specifications
For the engineer who thinks in specs, here is the breakdown of the standard architecture:
| Feature | Specification | The "Why" |
|---|---|---|
| Volume Range | 30ml – 1000ml | Matches the scale of reaction (Analytical vs. Bulk). |
| Primary Ports | 3 x Φ6.2mm | Accommodates standard Working, Counter, and Reference electrodes. |
| Gas Ports | 2 x Φ3.2mm | (Sealed units only) Enables inert gas purging and venting. |
| Jacket | Double-layer | Provides isothermal stability via water bath circulation. |
Conclusion
In experimental science, you cannot control what you cannot measure. But more importantly, you cannot measure accurately what you cannot stabilize.
The double-layer electrolytic cell is a vessel, but its function is insurance. It ensures that when you see a spike in your data, it’s because of your chemistry, not because the AC turned off in the lab.
At KINTEK, we understand that great research is built on reliable infrastructure. We provide high-precision electrolytic cells designed to eliminate the variables that keep you up at night.
Ready to stabilize your system? Contact Our Experts to find the exact configuration for your research needs.
Visual Guide
Related Products
- Double-Layer Water Bath Electrolytic Electrochemical Cell
- H-Type Double-Layer Optical Electrolytic Electrochemical Cell with Water Bath
- Double Layer Five-Port Water Bath Electrolytic Electrochemical Cell
- Multifunctional Electrolytic Electrochemical Cell Water Bath Single Layer Double Layer
- PTFE Electrolytic Cell Electrochemical Cell Corrosion-Resistant Sealed and Non-Sealed
Related Articles
- The Thermodynamics of Precision: Mastering the Double-Layer Electrolytic Cell
- The Quiet Discipline: Mastering the Post-Use Protocol for Five-Port Electrolytic Cells
- Exploring the Multifunctional Electrolytic Cell Water Bath: Applications and Benefits
- The Thermodynamics of Consistency: Mastering the Invisible Variable in Electrolysis
- The Art of the Shutdown: Engineering Reliability in Electrochemical Cells




















