The primary instrument used in modern IR spectroscopy is the Fourier Transform Infrared (FTIR) Spectrometer. This device rapidly acquires high-resolution spectral data by analyzing how a material absorbs infrared light. While older dispersive instruments exist, the FTIR spectrometer is the current standard due to its superior speed, sensitivity, and signal-to-noise ratio.
The core of modern infrared analysis isn't just a prism or grating, but a sophisticated system built around an interferometer. Understanding how this component works is key to understanding why FTIR has become the dominant method for chemical identification.

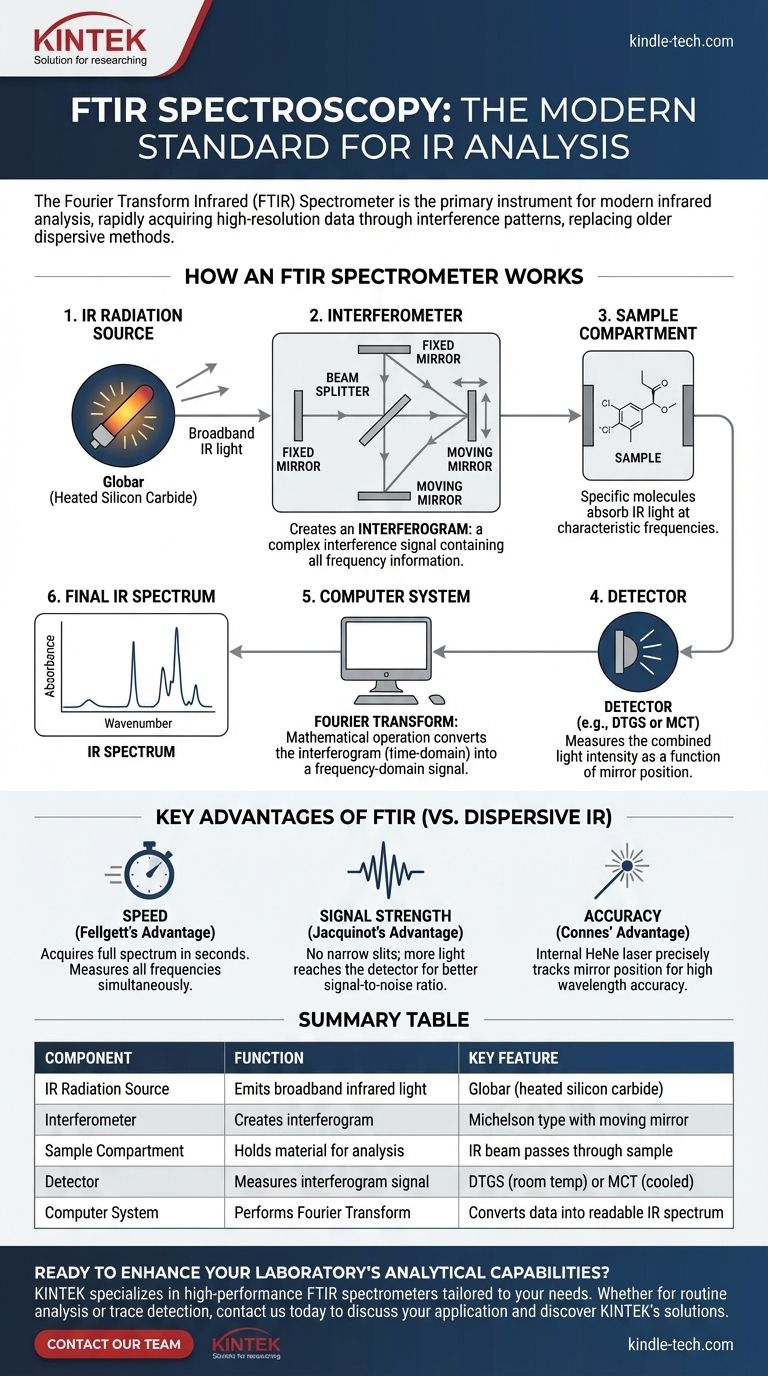
How an FTIR Spectrometer Works
At the heart of every FTIR spectrometer is a component that older instruments lack: an interferometer. The most common type is the Michelson interferometer.
The Role of the Interferometer
An interferometer's job is to split a beam of infrared light from the source into two separate beams. One beam travels a fixed distance to a stationary mirror and reflects back. The other travels to a moving mirror, which continuously alters the beam's path length before it also reflects back.
When these two beams are recombined, they "interfere" with each other either constructively (creating a stronger signal) or destructively (canceling each other out). This interference pattern changes as the moving mirror scans back and forth.
From Interferogram to Spectrum
The detector does not measure a spectrum directly. Instead, it measures the combined light intensity as a function of the moving mirror's position. This resulting signal is called an interferogram.
The interferogram is a complex signal that contains all the necessary frequency information simultaneously. A computer then performs a mathematical operation called a Fourier Transform on this interferogram. This calculation effectively decodes the interference pattern, converting it from a time-domain signal (intensity vs. mirror position) into a frequency-domain signal (intensity vs. wavenumber).
The final output is the familiar IR spectrum, a plot showing which frequencies of infrared light were absorbed by the sample.
The Key Components of an FTIR System
An FTIR spectrometer is a system of integrated parts, each with a specific function.
1. IR Radiation Source
The system requires a source that emits continuous, broadband infrared radiation. Common sources include a Globar (a silicon carbide rod heated to over 1000°C) or other ceramic filaments that glow when heated.
2. Interferometer
As discussed, this is the central component, typically a Michelson interferometer with a beam splitter, a fixed mirror, and a moving mirror. It's responsible for modulating the IR signal to produce the interferogram.
3. Sample Compartment
This is where the material being analyzed is placed. The IR beam passes through the sample, and specific functional groups within the molecules absorb light at their characteristic frequencies.
4. Detector
The detector measures the interferogram signal after it has passed through the sample. The most common type is a pyroelectric detector, such as deuterated triglycine sulfate (DTGS), which is reliable and operates at room temperature. For higher sensitivity or faster measurements, a mercury cadmium telluride (MCT) detector, which requires liquid nitrogen cooling, is used.
5. Computer System
A dedicated computer is essential. It controls the movement of the mirror, collects the data from the detector, performs the Fourier Transform, and displays the final spectrum for the analyst.
Understanding the Trade-offs: FTIR vs. Dispersive IR
Before FTIR became dominant, chemists used dispersive infrared spectrometers. Understanding the difference highlights why FTIR is the modern standard.
Dispersive Spectrometers (The Old Way)
A dispersive instrument uses a monochromator, such as a prism or a diffraction grating, to physically separate the infrared light into its component frequencies. It then scans through these frequencies one at a time, slowly measuring the absorption at each point to build the spectrum.
The Advantages of FTIR
FTIR spectrometers have three main advantages, collectively known as Fellgett's, Jacquinot's, and Connes' advantages.
- Speed (Fellgett's Advantage): Because the FTIR measures all frequencies simultaneously rather than one by one, it can acquire a full spectrum in a second or less. A dispersive instrument might take several minutes.
- Signal Strength (Jacquinot's Advantage): FTIR systems do not require narrow slits like dispersive instruments do to achieve resolution. This allows significantly more light (energy) to reach the detector, resulting in a much stronger signal and better signal-to-noise ratio.
- Accuracy (Connes' Advantage): The use of a HeNe laser to precisely track the moving mirror's position provides extremely high wavelength accuracy and precision, making spectra highly reproducible.
Making the Right Choice for Your Analysis
While the FTIR spectrometer is the standard instrument, the specific configuration depends on your analytical needs.
- If your primary focus is routine quality control or teaching: A standard benchtop FTIR with a room-temperature DTGS detector is robust, reliable, and cost-effective.
- If your primary focus is trace analysis or fast kinetics: You need a high-performance FTIR equipped with a liquid nitrogen-cooled MCT detector for its superior sensitivity and speed.
- If your primary focus is analyzing difficult or opaque samples: You will need to pair the FTIR with a specialized sampling accessory, such as an Attenuated Total Reflectance (ATR) crystal.
Ultimately, the FTIR spectrometer is the definitive instrument for modern infrared analysis, offering unparalleled performance and versatility.
Summary Table:
| Component | Function | Key Feature |
|---|---|---|
| IR Radiation Source | Emits broadband infrared light | Globar (heated silicon carbide) |
| Interferometer | Splits and recombines light to create an interferogram | Michelson type with moving mirror |
| Sample Compartment | Holds the material being analyzed | IR beam passes through the sample |
| Detector | Measures the interferogram signal | DTGS (room temp) or MCT (cooled, high sensitivity) |
| Computer System | Performs Fourier Transform and displays spectrum | Converts data into readable IR spectrum |
Ready to enhance your laboratory's analytical capabilities?
KINTEK specializes in providing high-performance FTIR spectrometers and lab equipment tailored to your specific research and quality control needs. Whether you require a robust benchtop model for routine analysis or a high-sensitivity system for trace detection, our experts can help you select the perfect instrument.
Contact our team today to discuss your application and discover how KINTEK's solutions can drive precision and efficiency in your laboratory.
Visual Guide
Related Products
- XRF & KBR plastic ring lab Powder Pellet Pressing Mold for FTIR
- XRF & KBR steel ring lab Powder Pellet Pressing Mold for FTIR
People Also Ask
- What is the XRF method of sampling? Achieve Accurate Elemental Analysis with Proper Sample Prep
- What is the purpose of using a mold for pellet pressing when preparing catalyst test samples? Ensure Data Consistency
- How are samples prepared for XRF analysis? Achieve Accurate and Reliable Results
- What is the primary function of a powder pellet press in the preparation of fillers? Achieve Superior Ceramic Joining
- What is the XRF pressed pellet method? A Fast, Cost-Effective Sample Prep Guide

