The quartz tube is the silent partner in your laboratory. It is transparent, seemingly inert, and capable of withstanding extreme thermal environments. Because of this resilience, it is often treated with a dangerous level of casualness.
But quartz is not just high-temp glass. It is a precision component composed of silicon dioxide (SiO₂), possessing a near-zero coefficient of thermal expansion. This specific molecular structure allows it to survive the furnace.
However, that structure is vulnerable.
Most catastrophic tube failures—cracks, shattering, or devitrification—are not caused by the heat itself. They are caused by what we leave behind inside the tube.
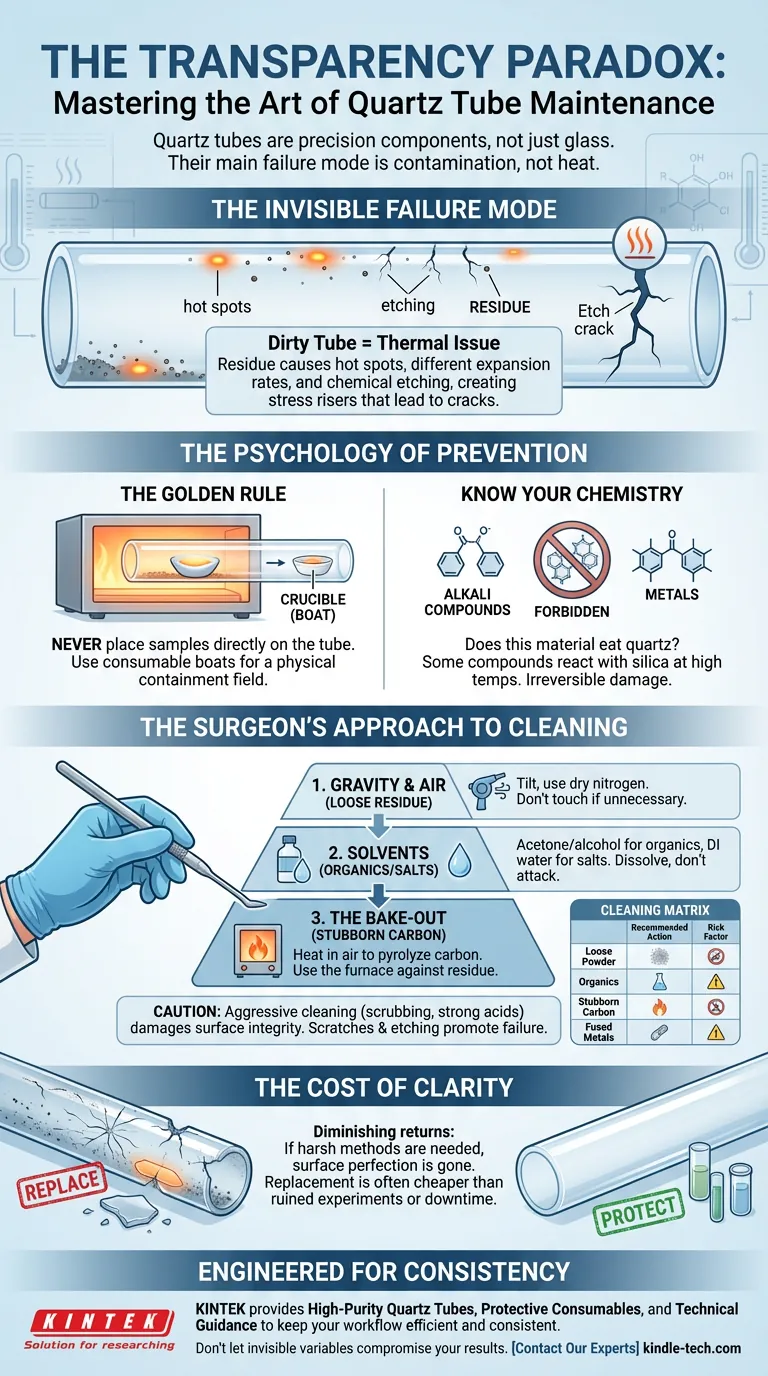
The Invisible Failure Mode
When a quartz tube is dirty, we tend to think of it as a hygiene issue. In reality, it is a thermal engineering issue.
Residue left on the tube wall changes the thermodynamics of the reaction chamber. A seemingly harmless smudge of carbon or a trace of metal oxide acts as a localized distinct material.
When the furnace ramps up:
- The residue expands at a different rate than the quartz.
- The residue absorbs heat differently, creating "hot spots."
- The residue reacts chemically with the silica, etching the surface.
This etching creates microscopic stress risers. Like a windshield with a tiny stone chip, the integrity of the entire vessel is compromised. The next time you ramp the temperature, that microscopic etch becomes a macroscopic crack.
Cleanliness is not about aesthetics. It is about preserving the structural physics of the tube.
The Psychology of Prevention
The most effective way to clean a quartz tube is to never let it get dirty in the first place.
This sounds obvious, yet it is the step most often skipped in the rush of experimentation. Morgan Housel often notes that "risk is what's left over when you think you've thought of everything." In lab work, risk is what touches the tube walls.
The Crucible Defense
The single greatest return on investment in a furnace lab is not the furnace itself, but the humble sample boat.
Never place samples directly on the tube. This is the golden rule.
By using high-quality ceramic or quartz crucibles (boats), you create a physical containment field. You are paying a few dollars for a consumable boat to protect a component worth hundreds or thousands.
Know Your Chemistry
Before a run, ask the hard question: Does this material eat quartz?
Alkali compounds and certain metals have a voracious appetite for SiO₂ at high temperatures. If you are heating materials that react with silica, no amount of post-run cleaning will save the tube. The damage is immediate and irreversible.
The Surgeon’s Approach to Cleaning
Sometimes, despite our best efforts, contamination happens. When it does, you must switch from a scientist to a surgeon. The goal is to remove the intruder without harming the patient.
Aggressive cleaning is often as damaging as the contaminant itself. Scrubbing creates scratches; scratches become cracks. Strong acids etch the glass; etching promotes devitrification.
Follow this hierarchy of intervention:
- Gravity and Air: If the residue is loose (soot or powder), tilt the tube or use dry nitrogen. Do not touch the surface if you don't have to.
- Solvents: For organics, use acetone or isopropyl alcohol. For salts, use deionized water. These dissolve the problem without attacking the quartz matrix.
- The Bake-Out: Use the furnace against the residue. Heating the empty tube in the presence of oxygen can pyrolyze carbon-based contaminants.
The Cleaning Matrix
| Contaminant Type | Recommended Action | The Risk Factor |
|---|---|---|
| Loose Powder | Mechanical Removal: Soft brush or tilt. | High risk of scratching if metal tools are used. |
| Organics | Solvent Wash: Acetone / Alcohol. | Minimal risk. Ensure total drying before heating. |
| Stubborn Carbon | Thermal Bake-out: Heat in air. | Effective, but requires time and energy. |
| Fused Metals | Dilute Acid: (Use extreme caution). | High risk. Acids can etch the quartz surface. |
The Cost of Clarity
There comes a point in the lifecycle of every piece of equipment where maintenance yields diminishing returns.
If a tube requires harsh acids (like hydrofluoric acid) or abrasive scrubbing to clean, its surface perfection is likely already gone. An etched tube is a ticking clock. It introduces variables into your thermal profile that you cannot control.
In these moments, the most scientific decision is replacement.
The cost of a new quartz tube is almost always lower than the cost of a ruined experiment, a lost sample, or the downtime caused by a tube shattering mid-cycle.
Engineered for Consistency
At KINTEK, we understand that the quartz tube is the heart of your furnace. We view laboratory equipment not as static objects, but as dynamic systems where material science meets thermal precision.
We provide the ecosystem required to protect that system:
- High-Purity Quartz Tubes: Manufactured for maximum thermal shock resistance.
- Protective Consumables: A wide range of alumina and quartz boats to prevent contamination before it starts.
- Technical Guidance: Expertise on material compatibility to keep your workflow efficient.
Don't let invisible variables compromise your results.
Visual Guide
Related Products
- Laboratory Rapid Thermal Processing (RTP) Quartz Tube Furnace
- 1200℃ Split Tube Furnace with Quartz Tube Laboratory Tubular Furnace
- Optical Window Glass Substrate Wafer Quartz Plate JGS1 JGS2 JGS3
- High Temperature Resistant Optical Quartz Glass Sheet
- Rotary Tube Furnace Split Multi Heating Zone Rotating Tube Furnace
Related Articles
- The Architecture of Heat: Choosing Between Control and Capacity
- The Architecture of Isolation: Anatomy of a Tube Furnace
- The Secrets To tube furnace
- From Crack to Complete: A Scientist's Guide to Eliminating Catastrophic Tube Furnace Failures
- Beyond Temperature: Mastering the Invisible Environment Inside a Tube Furnace














