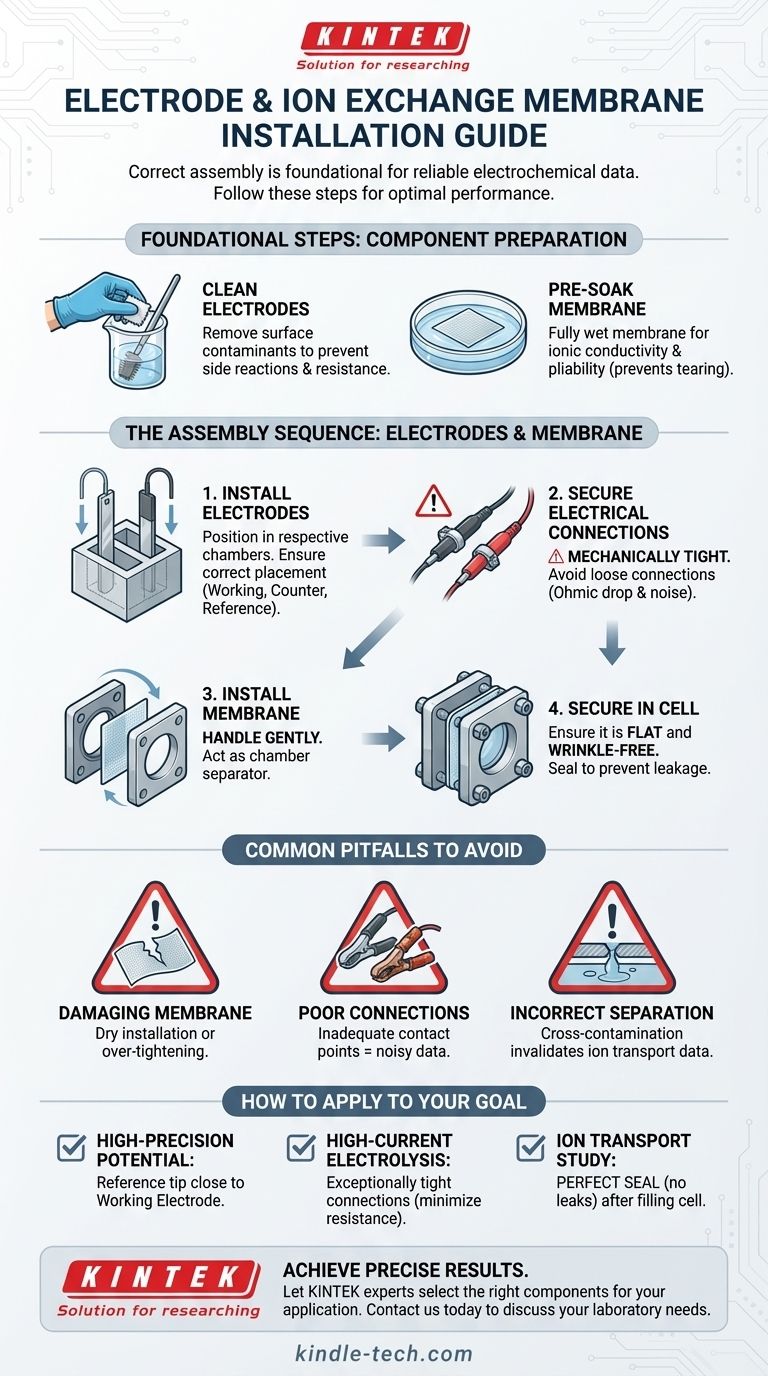
To correctly install electrodes and an ion exchange membrane, the procedure begins with component preparation. You must first install the cleaned electrodes into the electrolytic cell, ensuring they are positioned correctly and have a tight electrical connection. Following this, the pre-soaked ion exchange membrane is carefully installed between the anode and cathode chambers to ensure a proper seal.
The success of your electrochemical experiment hinges not just on the assembly sequence, but on the integrity of each component. Proper preparation and careful installation are critical for ensuring accurate ion transport and reliable electrical connections, which form the foundation of valid data.

Foundational Steps: Component Preparation
Before any assembly begins, preparing your components correctly is the most critical step. Skipping this stage can compromise your entire experiment.
Cleaning the Electrodes
Electrodes must be scrupulously clean. Any surface contaminants can introduce unwanted side reactions or increase electrical resistance, skewing your results. Follow the specific cleaning protocol for your electrode material.
Preparing the Ion Exchange Membrane
The membrane is the heart of chamber separation. It is crucial to pre-soak the membrane in the electrolyte solution you will be using. This ensures it is fully wetted, which dramatically improves its ionic conductivity and makes it more pliable, preventing tearing during installation.
The Assembly Sequence: Electrodes and Membrane
A logical assembly order prevents damage and ensures correct positioning. The standard practice is to install the electrodes first, followed by the delicate membrane.
Installing the Electrodes
Carefully insert the cleaned electrodes into their respective ports in the electrolytic cell. Position them according to your experimental design.
Ensuring a Tight Electrical Connection
The connection between your potentiostat leads and the electrodes must be mechanically tight and reliable. A loose connection introduces noise and unwanted resistance (Ohmic drop), which can corrupt your voltage and current measurements.
Correct Electrode Positioning
Typically, the working electrode and counter electrode are placed in separate chambers (the anode and cathode compartments). The reference electrode, if used, must be placed in the same chamber as the working electrode to accurately measure its potential.
Installing the Membrane
With the electrodes in place, you can now install the membrane. Handle the pre-soaked membrane gently, as it can be fragile.
Securing the Membrane in the Cell
Place the membrane between the two halves of your electrolytic cell. A small amount of electrolyte can act as a lubricant to help it slide into place without damage. Ensure it is flat and free of wrinkles before sealing the cell.
Common Pitfalls to Avoid
Even with the correct procedure, small mistakes can lead to failed experiments. Being aware of these common pitfalls is essential for achieving reliable and repeatable results.
Pitfall: Damaging the Membrane
Installing a membrane while it is dry is a common cause of tearing. Always pre-soak it. Additionally, over-tightening the cell can crush or puncture the membrane, creating a leak.
Pitfall: Poor Electrical Connections
Relying on simple alligator clips without ensuring a firm, clean contact point is a frequent source of error. This leads to noisy data and inaccurate potential control.
Pitfall: Incorrect Chamber Separation
If the membrane is not properly sealed, electrolyte will leak between the anode and cathode chambers. This cross-contamination will invalidate any results related to ion transport or separated electrochemical reactions.
How to Apply This to Your Goal
Final checks should be aligned with your specific experimental objective.
- If your primary focus is high-precision potential measurement: Double-check that the tip of your reference electrode is positioned as close as possible to the surface of the working electrode.
- If your primary focus is high-current electrolysis: Confirm that your electrode connections are exceptionally tight to minimize resistive heating and voltage loss.
- If your primary focus is studying ion transport: Ensure the membrane is perfectly sealed with no visible leaks after filling the cell with electrolyte.
A methodical and careful setup is your first and most important step toward generating trustworthy scientific data.
Summary Table:
| Step | Key Action | Purpose |
|---|---|---|
| 1. Preparation | Clean electrodes; Pre-soak membrane | Remove contaminants; Ensure ionic conductivity & prevent damage |
| 2. Electrode Installation | Insert electrodes into cell; Secure electrical connections | Establish reliable current path; Minimize Ohmic drop |
| 3. Membrane Installation | Place pre-soaked membrane between cell halves; Ensure proper seal | Separate chambers; Prevent electrolyte cross-contamination |
| 4. Final Check | Verify connections and seal based on experimental goal (e.g., precision measurement, high current) | Ensure setup aligns with specific research objectives |
Achieve precise and reliable electrochemical results with confidence. Proper cell assembly is foundational to your research success. KINTEK specializes in high-quality lab equipment and consumables, including durable electrolytic cells, electrodes, and ion exchange membranes designed for exceptional performance and longevity. Let our experts help you select the right components for your specific application. Contact us today to discuss your laboratory needs and ensure your experiments are built on a solid foundation.
Visual Guide
Related Products
- Proton Exchange Membrane for Batteries Lab Applications
- Anion Exchange Membrane for Laboratory Use
- Customizable PEM Electrolysis Cells for Diverse Research Applications
- Polyethylene Separator for Lithium Battery
- Conductive Carbon Cloth Carbon Paper Carbon Felt for Electrodes and Batteries
People Also Ask
- What is a common application for proton exchange membranes in laboratory setups? Ensuring Precise Electrochemical Analysis
- What is the function of a PEM in an MFC? Optimize Proton Migration & Power Efficiency
- What initial steps are required before using a new proton exchange membrane? Ensure Peak Performance and Longevity
- What is a proton exchange membrane? The Selective Heart of Hydrogen Energy Systems
- How can the performance and condition of a proton exchange membrane be monitored? A Guide to PEM Health & Longevity







