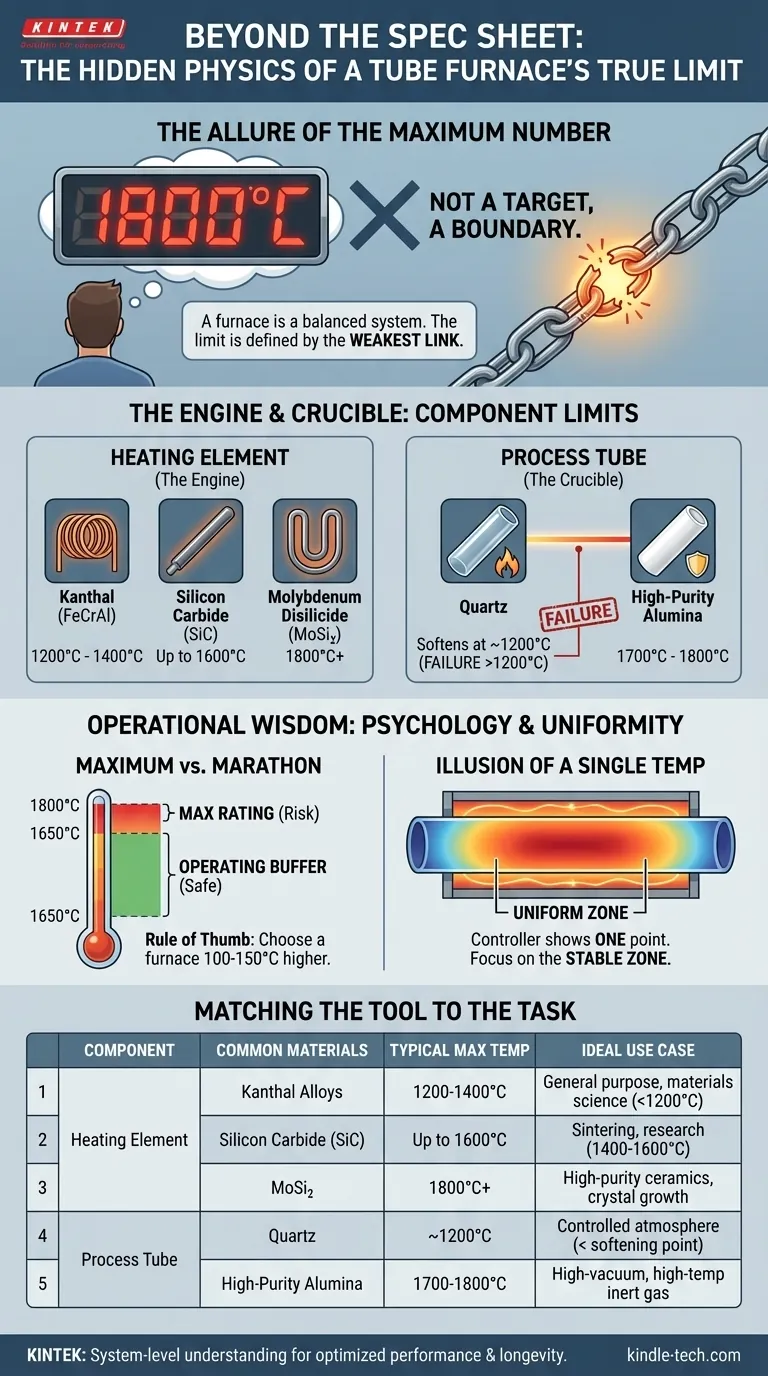
The Allure of the Maximum Number
When evaluating a high-temperature tube furnace, the first question is almost always the same: "How hot can it get?"
It’s a natural human impulse. We are drawn to extremes, to the highest number on the spec sheet. We see 1800°C and assume it’s inherently better than 1200°C. But this simple question hides a complex reality and often leads to poor decisions, premature equipment failure, and compromised research.
A tube furnace isn't a simple appliance; it's a balanced system of interacting components under extreme stress. The "maximum temperature" is not a target to be aimed for, but a boundary defined by the single weakest link in that system.
The right question is not "how hot can it get?" but rather, "what limits its temperature, and how do those limits align with my work?"
A System Defined by Its Weakest Link
To understand a furnace’s true capabilities, you have to look past the single number and see the components that produce and contain the heat. The maximum temperature is a promise made by the entire system, but it can be broken by any single part.
The Engine of Heat: The Heating Element
The heart of the furnace is its heating element. The material used here is the primary determinant of the furnace's thermal potential. Each has a hard physical limit beyond which it will rapidly degrade and fail.
- Kanthal (FeCrAl) Alloys: The workhorse for general applications, reliably operating up to 1200°C - 1400°C. Cost-effective and durable within their range.
- Silicon Carbide (SiC): The next step up, essential for processes requiring sustained heat up to 1600°C.
- Molybdenum Disilicide (MoSi₂): The choice for extreme temperatures, engineered to perform consistently at 1800°C and even higher in specialized designs.
The Crucible of Containment: The Process Tube
While the element creates the heat, the process tube contains the experiment. It must endure the same extreme temperatures while maintaining its structural integrity and protecting the sample. Often, the tube itself becomes the limiting factor.
- Quartz Tubes: An excellent and common material, but it has a non-negotiable ceiling. Above approximately 1200°C, quartz begins to soften and deform.
- High-Purity Alumina Tubes: For any work above 1200°C, these ceramic tubes are mandatory. They can withstand temperatures from 1700°C to 1800°C, making them the partner for SiC and MoSi₂ elements.
Pushing a quartz tube into a 1400°C furnace is not a matter of risk; it is a guarantee of failure.
The Psychology of Operational Limits
Understanding the physics is only half the battle. The other half is fighting the psychological urge to push the equipment to its absolute limit—a behavior that is both costly and counterproductive.
Maximum vs. Marathon Temperature
A furnace rated for 1800°C is not designed to be run at 1800°C every day. This "maximum temperature" is a capability, not a recommended cruising speed.
Operating any furnace at its absolute peak drastically shortens the lifespan of its heating elements and places immense thermal stress on its insulation and structure. A wise rule of thumb is to choose a furnace with a maximum rating at least 100-150°C higher than your typical operating temperature. This buffer is the difference between an instrument that lasts for years and one that fails in months.
The Illusion of a Single Temperature
The number on the digital controller represents the temperature at a single point—the tip of the thermocouple, typically in the center of the heating zone. It does not represent the temperature throughout the entire tube.
Temperature naturally drops off toward the ends of the tube. For processes like crystal growth or annealing semiconductor wafers, the length of the stable and consistent "uniform zone" is far more critical than a peak number. Chasing the maximum temperature can blind you to the more important metric of thermal uniformity.
Matching the Tool to the Task
Selecting the right furnace is not about buying the most powerful one. It's about precisely matching the equipment's systemic capabilities to your scientific goals. The choice becomes clear when you define your needs first.
| Component | Common Materials | Typical Max Temperature | Ideal Use Case |
|---|---|---|---|
| Heating Element | Kanthal Alloys | 1200°C - 1400°C | General purpose, materials science, annealing below 1200°C |
| Silicon Carbide (SiC) | Up to 1600°C | Sintering, research requiring 1400°C - 1600°C | |
| Molybdenum Disilicide (MoSi₂) | 1800°C+ | High-purity ceramics, crystal growth, extreme temp R&D | |
| Process Tube | Quartz | ~1200°C | Controlled atmosphere work below the softening point |
| High-Purity Alumina | 1700°C - 1800°C | High-vacuum and high-temperature inert gas processes |
Choosing the right system requires more than just reading a spec sheet; it requires expertise in how these components interact under the stress of your specific application. At KINTEK, we provide not just the equipment, but the system-level understanding to ensure you get the right tool for your research, optimized for performance and longevity.
If you need to navigate these critical trade-offs, Contact Our Experts.
Visual Guide
Related Products
- Rotary Tube Furnace Split Multi Heating Zone Rotating Tube Furnace
- Laboratory Vacuum Tilt Rotary Tube Furnace Rotating Tube Furnace
- 1200℃ Split Tube Furnace with Quartz Tube Laboratory Tubular Furnace
- 1400℃ Laboratory High Temperature Tube Furnace with Alumina Tube
- 1700℃ Laboratory High Temperature Tube Furnace with Alumina Tube
Related Articles
- Rotary Furnaces: A Comprehensive Guide to Advanced Materials Processing
- Exploring the Advantages of Rotary Tube Furnaces in Industrial and Laboratory Applications
- The Geometry of Heat: Engineering the Perfect Thermal Environment
- Exploring the Benefits and Applications of Rotary Furnaces: A Comprehensive Guide
- Unlocking Efficiency: The Ultimate Guide to Rotary Tube Furnaces




















