The Psychology of the "Perfect" Environment
In the chaotic world of material science, variables are the enemy.
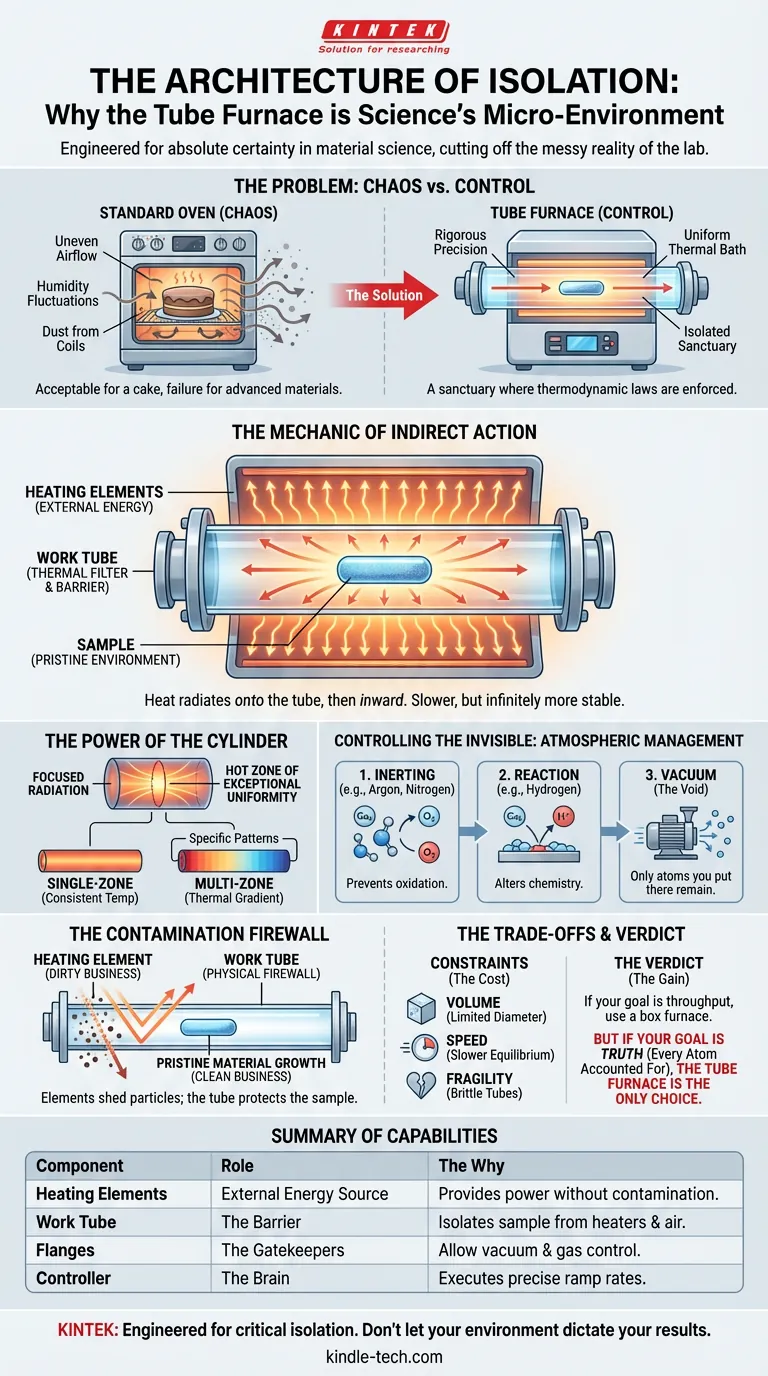
If you bake a cake in a standard oven, you accept a certain amount of chaos. Uneven airflow, fluctuations in humidity, and the microscopic dust shedding from the heating coils are all part of the process. For a cake, this is character.
For advanced semiconductors or nanomaterials, this is failure.
The tube furnace exists to solve a psychological and engineering problem: the need for absolute certainty. It is not merely a device that gets things hot. It is a machine designed to create a "micro-environment"—a sanctuary where the laws of thermodynamics are enforced with rigorous precision, cut off from the messy reality of the outside lab.
Here is how that isolation is engineered.
The Mechanic of Indirect Action
The fundamental difference between a standard box furnace and a tube furnace is the relationship between the heater and the heated.
In most ovens, the sample sits in the same room as the fire. In a tube furnace, they are separated by a wall.
The Heating Elements
Surrounding the central chamber are high-resistance heating elements. When powered, they glow with intense energy. However, they remain external. They never touch the atmosphere where your chemistry happens.
The Work Tube
Running through the center of this inferno is the work tube. Crafted from quartz, alumina, or corundum, this tube is the hero of the system. It acts as a thermal filter.
The Transfer
The heat does not blast the sample. It radiates onto the exterior of the tube, soaks through the ceramic or glass wall, and then radiates inward toward the center axis.
This indirect heating method is slower, yes. But it is infinitely more stable. It smooths out the spikes. It turns a violent energy source into a gentle, uniform bath of thermal radiation.
The Power of the Cylinder
Why a tube? Why not a box?
Geometry dictates performance. A cylinder is the most efficient shape for focused radiation.
When heat radiates from the walls of a cylinder, it converges on the central axis. This creates a "hot zone" of exceptional uniformity.
Single vs. Multi-Zone
- Single-Zone: Ideal for tasks requiring one consistent temperature throughout the sample.
- Multi-Zone: These furnaces use independent controllers along the length of the tube. This allows you to create a specific thermal gradient—hot at one end, cool at the other—forcing materials to migrate or crystallize in specific patterns.
Controlling the Invisible: Atmospheric Management
The true "engineer’s romance" of the tube furnace is not the heat. It is the vacuum.
Because the work tube is a continuous, non-porous vessel, you can attach sealing flanges to both ends. This transforms the furnace from a heater into a chemical reactor.
The Three States of Control
- Inerting: You can purge the tube with Argon or Nitrogen. This prevents oxidation, allowing metals to anneal without rusting.
- Reaction: You can introduce reactive gases (like Hydrogen) to strip oxygen away or alter the surface chemistry of a sample.
- Vacuum: You can pull the air out entirely. In a high-vacuum tube furnace, you are processing materials in a void, ensuring that the only atoms present are the ones you put there.
The Contamination Firewall
In high-purity synthesis, the heating element itself is a contaminant. Molybdenum disilicide or resistance wire can shed microscopic particles as they age.
If your sample is in a box furnace, those particles land on your work.
In a tube furnace, the work tube acts as a physical firewall. The dirty business of generating heat happens on the outside; the pristine business of material growth happens on the inside.
The Trade-offs: When to Choose the Tube
Everything in engineering is a trade-off. You pay for precision with capacity.
The Constraints:
- Volume: You are limited by the diameter of the tube. You cannot process large bulk parts.
- Speed: Indirect heating takes longer to reach equilibrium than direct heating.
- Fragility: High-temperature ceramic tubes are brittle. They require respect and careful handling.
The Verdict: If your goal is throughput—baking 500 ceramic cups—use a box furnace.
But if your goal is truth—synthesizing a material where every atom must be accounted for—the tube furnace is the only logical choice.
Summary of Capabilities
| Component | Role | The "Why" |
|---|---|---|
| Heating Elements | External Energy Source | Provides power without contamination risk. |
| Work Tube | The Barrier | Isolates the sample from the heaters and the lab air. |
| Flanges | The Gatekeepers | Allow for vacuum sealing or gas flow control. |
| Controller | The Brain | Executes precise ramp rates to prevent thermal shock. |
Conclusion
The tube furnace is a testament to the idea that great science requires boundaries. By isolating the sample, we gain control over it.
At KINTEK, we understand that your research hinges on the integrity of that micro-environment. Our tube furnaces are engineered to provide that critical isolation, whether you require high-vacuum capabilities or multi-zone thermal gradients.
Don't let your environment dictate your results.
Visual Guide
Related Products
- Rotary Tube Furnace Split Multi Heating Zone Rotating Tube Furnace
- Laboratory Vacuum Tilt Rotary Tube Furnace Rotating Tube Furnace
- 1200℃ Split Tube Furnace with Quartz Tube Laboratory Tubular Furnace
- 1400℃ Laboratory High Temperature Tube Furnace with Alumina Tube
- 1700℃ Laboratory High Temperature Tube Furnace with Alumina Tube
Related Articles
- The Geometry of Heat: Engineering the Perfect Thermal Environment
- Rotary Furnaces: A Comprehensive Guide to Advanced Materials Processing
- Exploring the Advantages of Rotary Tube Furnaces in Industrial and Laboratory Applications
- Exploring the Functions and Advantages of Rotary Tube Furnaces
- Unlocking Efficiency: Exploring the Advantages and Applications of Rotary Tube Furnaces




















