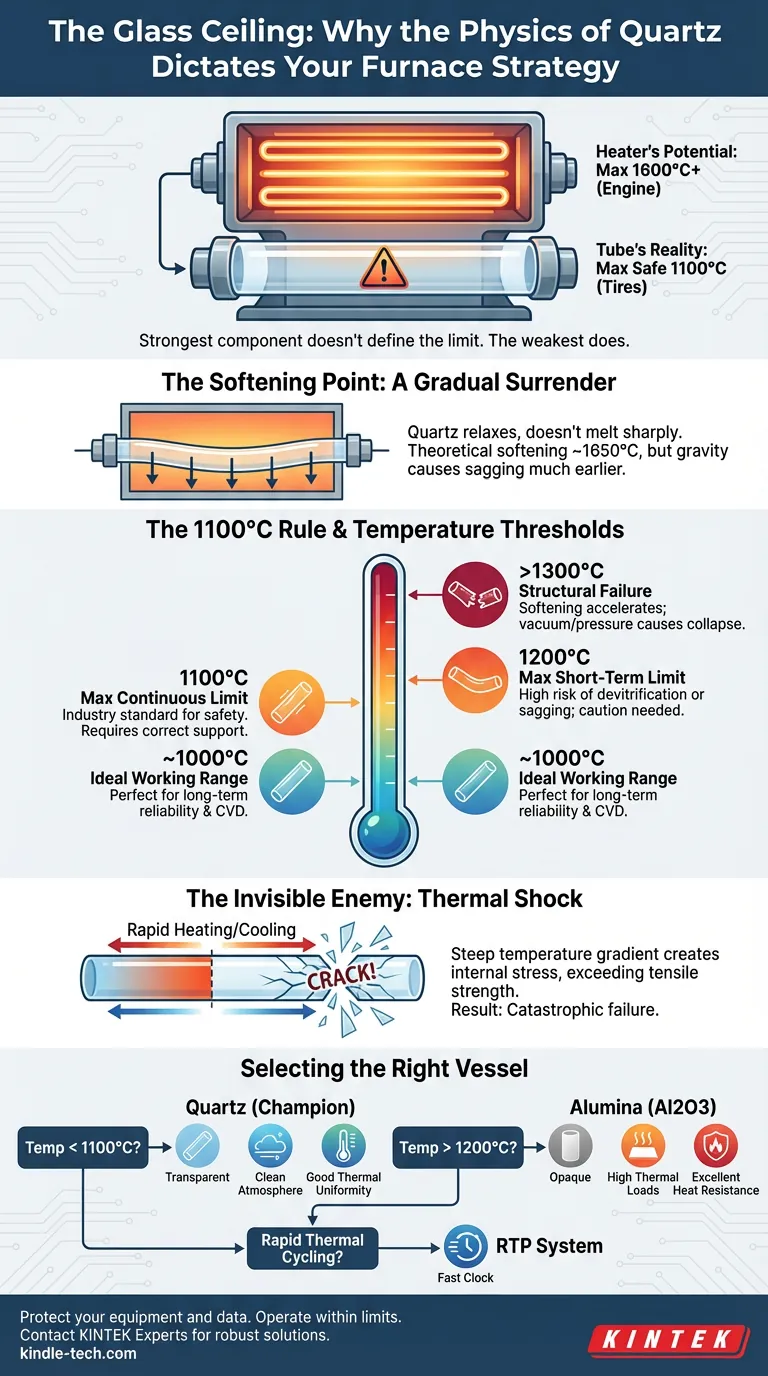
We tend to judge a machine by its maximum capacity. In the automotive world, we look at top speed. In the laboratory, we look at the maximum temperature rating on the furnace controller.
But in high-temperature thermodynamics, the strongest component does not define the system's limit. The weakest one does.
When you operate a high-temperature tube furnace, the heating elements—often made of Molybdenum Disilicide or Silicon Carbide—are the engine. They are capable of roaring up to 1600°C or higher without breaking a sweat.
However, the quartz tube is the tires.
No matter how powerful the engine is, if you push the tires beyond their physical grip, you crash. Understanding this discrepancy between the heater's potential and the tube's reality is the difference between a successful experiment and a catastrophic, glass-shattering failure.
The Softening Point: A Gradual Surrender
Unlike metals, which often hold their rigid structure right up until they reach a sharp melting point, glass behaves differently.
Quartz is a liquid that has forgotten how to flow. As you heat it, it doesn't suddenly melt; it relaxes.
High-purity fused quartz has a theoretical softening point of approximately 1650°C (3000°F). In a vacuum of perfect physics, this is where the structure begins to lose its rigidity.
But your lab is not a vacuum of perfect physics.
Gravity is relentless. At temperatures approaching this softening point, a quartz tube—spanning the length of a furnace and potentially loaded with samples—will begin to sag under its own weight. This deformation is not just cosmetic; it compromises the gas flow, the thermal uniformity, and the safety of the vessel.
The 1100°C Rule
Because of the risk of deformation, the "safe" operating ceiling is significantly lower than the material's theoretical limit.
For most laboratory applications, the line in the sand is 1100°C (2012°F).
- At 1000°C: Quartz is in its prime. It is robust, chemically inert, and thermally stable. This is the ideal zone for annealing and Chemical Vapor Deposition (CVD).
- At 1100°C: This is the practical continuous limit. You are safe here, provided the tube is clean and supported correctly.
- At 1200°C: You are pushing the envelope. This is possible for short durations under strictly controlled conditions, but the lifespan of the tube begins to degrade rapidly.
Temperature Thresholds at a Glance
| Parameter | Temperature | Operational Reality |
|---|---|---|
| Ideal Working Range | ~1000°C | Perfect for long-term reliability and CVD processes. |
| Max Continuous Limit | 1100°C | The industry standard for safety. |
| Max Short-Term Limit | 1200°C | High risk of devitrification or sagging; requires caution. |
| Structural Failure | >1300°C | Softening accelerates; vacuum/pressure causes collapse. |
The Invisible Enemy: Thermal Shock
Heat is not the only variable. The speed of heat is equally critical.
Quartz is beloved by engineers for its incredibly low coefficient of thermal expansion. It changes shape very little when heated. However, it is not invincible.
The most common way to destroy a quartz tube is not by melting it, but by startling it.
Thermal shock occurs when the temperature gradient across the glass wall is too steep—heating or cooling too rapidly. This creates internal stress that exceeds the tensile strength of the material. The result is a crack that snaps through the tube with the sound of a pistol shot.
If you are running a vacuum or positive pressure inside the tube, this stress is magnified. As the quartz softens near 1100°C, the pressure differential acts like a physical weight, pushing the walls inward or outward until failure occurs.
Selecting the Right Vessel
Engineering is the art of trade-offs. The quartz tube is an elegant solution—transparent, pure, and affordable—but it demands respect.
If your research requires:
- Temperatures below 1100°C: Quartz is the undisputed champion. It offers a clean atmosphere and excellent thermal uniformity.
- Temperatures exceeding 1200°C: You must abandon quartz. You need a ceramic material like Alumina (Al2O3), which can withstand much higher thermal loads but lacks the transparency and thermal shock resistance of quartz.
- Rapid thermal cycling: A standard quartz tube furnace may not be the right tool. You need a system designed for rapid thermal processing (RTP) or you must accept strict ramp-rate limitations.
Conclusion
The romance of the laboratory lies in pushing boundaries, but the machinery that makes discovery possible is governed by immutable laws.
A quartz tube furnace is a precise instrument, not a blunt force tool. By operating within the 1100°C limit and respecting the ramp rates, you protect both the integrity of your equipment and the validity of your data.
At KINTEK, we understand that your equipment is the foundation of your research. We provide robust laboratory solutions designed to handle the realities of high-temperature physics.
Whether you need high-purity quartz for sensitive CVD work or robust alumina for extreme heat, we can help you configure the right system.
Visual Guide
Related Products
- Laboratory Rapid Thermal Processing (RTP) Quartz Tube Furnace
- 1200℃ Split Tube Furnace with Quartz Tube Laboratory Tubular Furnace
- 1400℃ Laboratory High Temperature Tube Furnace with Alumina Tube
- Rotary Tube Furnace Split Multi Heating Zone Rotating Tube Furnace
- 1700℃ Laboratory High Temperature Tube Furnace with Alumina Tube
Related Articles
- Beyond Temperature: Mastering the Invisible Environment Inside a Tube Furnace
- The Secrets To tube furnace
- The Silent Partner in Pyrolysis: Engineering the Perfect Thermal Boundary
- From Crack to Complete: A Scientist's Guide to Eliminating Catastrophic Tube Furnace Failures
- The Thermal Divide: Navigating the Choice Between Tube and Chamber Furnaces




















