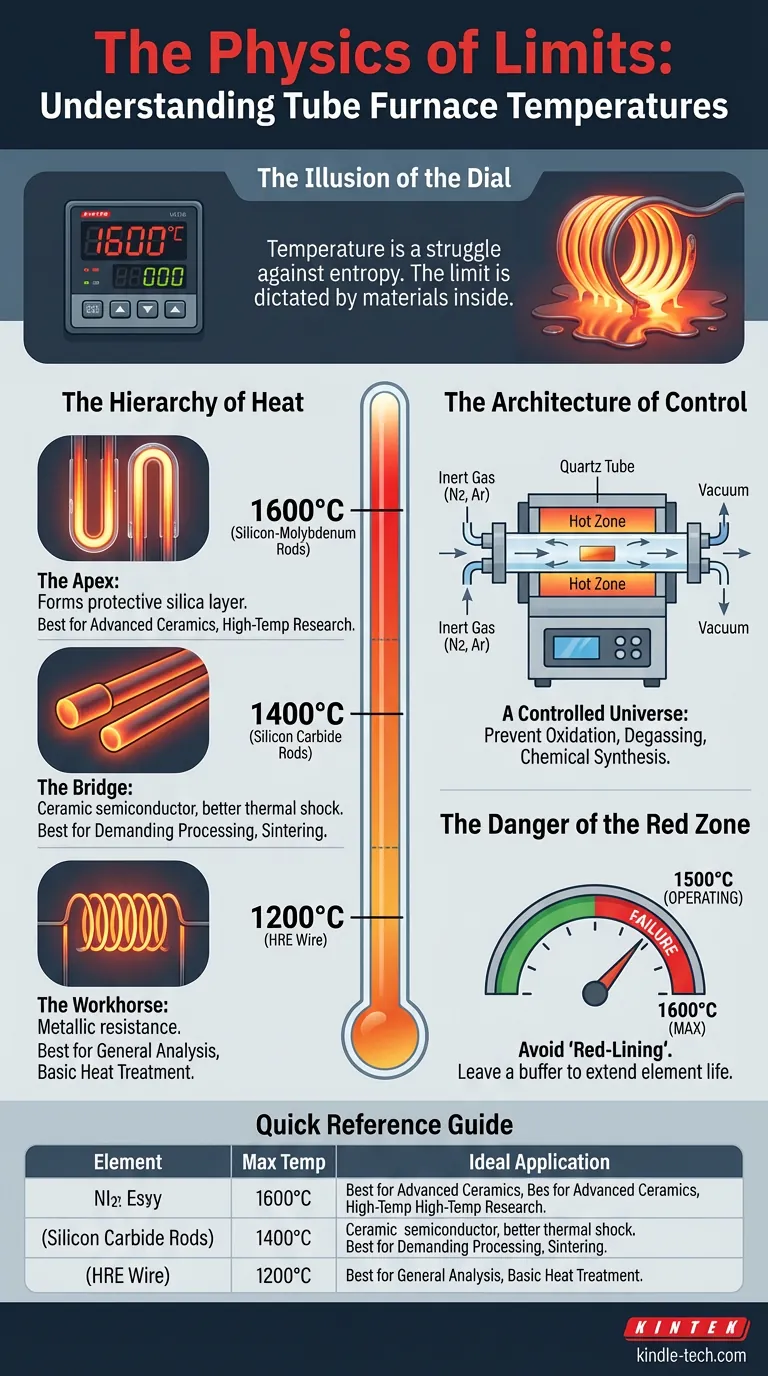
The Illusion of the Dial
In the laboratory, there is a seduction to high numbers. We look at a furnace specification sheet, see "1600°C," and assume that is the machine's natural state.
But temperature is not a static feature. It is a struggle against entropy.
A tube furnace is a vessel designed to contain a violent amount of energy in a very small space. The limit of that energy is not dictated by the controller on the front panel. It is dictated by the materials inside the chamber.
When you ask, "What is the temperature of a tube furnace?", you are actually asking a materials science question: What is the breaking point of the heating element?
Understanding this distinction is the difference between a successful experiment and a melted, expensive pile of scrap.
The Hierarchy of Heat
Engineering is the art of trade-offs. To achieve higher temperatures, we must trade common materials for exotic ones.
The thermal capacity of a tube furnace is determined strictly by the composition of its heating elements. There are three distinct tiers, each acting as a gatekeeper to a specific thermal realm.
1. The Workhorse: HRE Resistance Wire (Up to 1200°C)
Think of this as the elevated version of the filament in a toaster, but engineered for precision. High-Resistance Element (HRE) wire is reliable and cost-effective.
- The Physics: It relies on metallic resistance to generate heat.
- The Limit: Beyond 1200°C, the metal structure begins to degrade rapidly.
- Best For: General chemical analysis, basic heat treatment.
2. The Bridge: Silicon Carbon Rods (Up to 1400°C)
When metal fails, we turn to ceramics. Silicon Carbide (SiC) is a robust semiconductor material that can withstand thermal shock better than wire.
- The Physics: These rods glow with an intense, rigid heat.
- The Limit: They push the boundary to 1400°C, allowing for processing that requires significant energy input.
- Best For: Demanding materials processing and sintering.
3. The Apex: Silicon-Molybdenum Rods (Up to 1600°C)
At 1600°C, steel is a liquid. To sustain this environment, we use Molybdenum Disilicide (MoSi2).
- The Physics: When heated, these rods form a protective glass-like layer of silica on their surface, preventing oxidation even at extreme temperatures.
- The Limit: This is the current standard for non-induction high-temperature research.
- Best For: Advanced ceramics sintering, high-temperature research.
The Architecture of Control
The "temperature" is only half the story. The romance of the tube furnace lies in what accompanies the heat: The Atmosphere.
A standard box furnace is often just a hot box of air. A tube furnace is a controlled universe.
By inserting a quartz or alumina tube through the heating elements, we create a barrier. Inside that tube, we can strip away the oxygen. We can introduce nitrogen, argon, or hydrogen.
This allows for chemistry that nature usually forbids:
- Prevention of Oxidation: Keeping metals pure while hot.
- Degassing: Using vacuum pressure to pull trapped gases out of a solid lattice.
- Chemical Synthesis: Encouraging reactions that only occur in inert environments.
The Danger of the "Red Zone"
There is a psychological trap in engineering called "Red-Lining." It is the tendency to run a machine at exactly its maximum rated capacity.
If you buy a furnace rated for 1600°C and run it at 1600°C every day, you are choosing failure.
The operational reality:
- Rated Temperature vs. Operating Temperature: A wise engineer leaves a buffer. Running a 1200°C furnace at 1100°C extends the life of the elements exponentially.
- The Gradient: Heat is not perfectly uniform. The center of the tube—the "Hot Zone"—is accurate. The ends, near the insulation plugs, are cooler. Your sample must be positioned with intent.
Selecting Your Instrument
Choosing a furnace is not about buying the highest number you can afford. It is about matching the tool to the problem.
If you are annealing copper, a Silicon-Molybdenum furnace is financial overkill. If you are sintering zirconia, an HRE wire furnace is physically incapable of the task.
Quick Reference Guide
| Heating Element | Max Temp | Ideal Application |
|---|---|---|
| HRE Wire | 1200°C | General analysis, basic annealing |
| Silicon Carbon | 1400°C | Harder materials, faster ramp rates |
| Silicon-Molybdenum | 1600°C | Advanced ceramics, sintering, research |
Conclusion
The tube furnace is a testament to our ability to control the fundamental forces of nature—heat and atmosphere—on a desktop scale. But it requires respect for the materials that make it possible.
At KINTEK, we don't just sell equipment; we engineer solutions for the precise thermal environments your research demands. Whether you need the reliability of a 1200°C system or the extreme capability of a 1600°C workstation, we ensure the physics works in your favor.
Visual Guide
Related Products
- Rotary Tube Furnace Split Multi Heating Zone Rotating Tube Furnace
- Laboratory Vacuum Tilt Rotary Tube Furnace Rotating Tube Furnace
- 1200℃ Split Tube Furnace with Quartz Tube Laboratory Tubular Furnace
- 1400℃ Laboratory High Temperature Tube Furnace with Alumina Tube
- 1700℃ Laboratory High Temperature Tube Furnace with Alumina Tube
Related Articles
- Rotary Furnaces: A Comprehensive Guide to Advanced Materials Processing
- Maximize Rotary Furnace Performance: Design Advancements for Efficient Material Processing
- The Geometry of Heat: Engineering the Perfect Thermal Environment
- Unlocking Efficiency: Exploring the Advantages and Applications of Rotary Tube Furnaces
- Exploring Rotary Tube Furnaces: A Comprehensive Guide




















