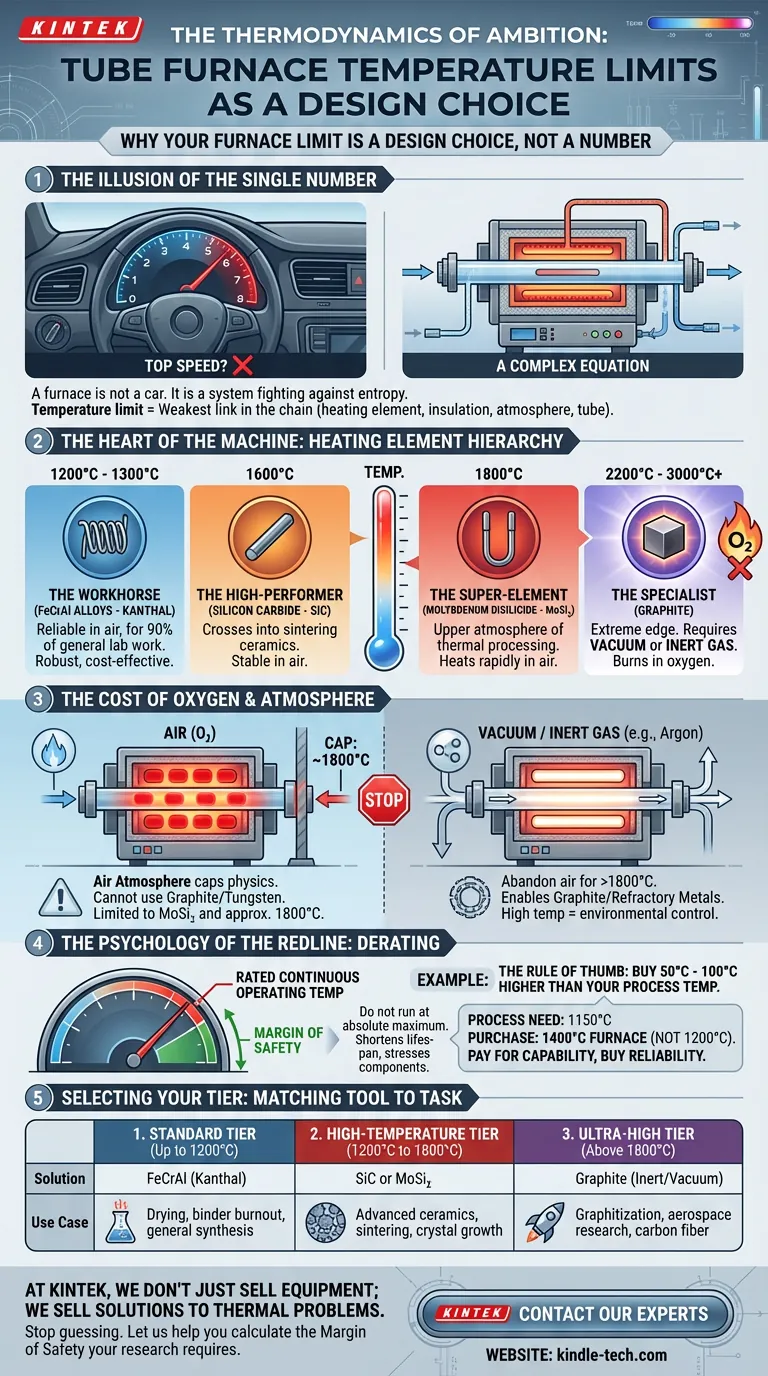
The Illusion of the Single Number
In the world of laboratory engineering, we often look for a single number to define capability. We ask, "What is the maximum temperature of this furnace?" as if asking for the top speed of a sports car.
But a furnace is not a car. It is a system fighting against entropy.
The answer to "how hot can it get?" is never a simple integer. It is a complex equation defined by the weakest link in the chain: the heating element, the insulation, the atmosphere, and the process tube itself.
A standard lab furnace might top out at 1200°C. A specialized graphite unit can scream past 3000°C.
The gap between those two numbers isn't just degrees. It is a fundamental difference in engineering philosophy and material science. To choose the right tool, you must understand the architecture of heat.
The Heart of the Machine: Heating Elements
The primary constraint on temperature is the material used to generate it.
When you push a material beyond its atomic limits, it doesn't just stop working. It oxidizes. It sags. It fails.
Here is the hierarchy of heat, defined by the elements that survive it:
- The Workhorse (FeCrAl Alloys): Known as Kanthal, these are the standard for 90% of general lab work. They are robust and inexpensive, operating reliably in air up to 1200°C – 1300°C.
- The High-Performer (Silicon Carbide): When you need to cross the threshold into sintering ceramics, SiC takes over. It remains stable in air up to 1600°C.
- The Super-Element (Molybdenum Disilicide): MoSi₂ elements are designed for the upper atmosphere of thermal processing. They heat rapidly and can operate continuously in air up to 1800°C.
- The Specialist (Graphite): This is the extreme edge. Graphite can reach 2200°C to 3000°C. But there is a catch: in the presence of oxygen, graphite burns. It requires a vacuum or inert gas to survive.
The Cost of Oxygen
This brings us to the most overlooked variable in thermal engineering: the atmosphere.
Heat does not exist in a vacuum—unless you build a machine specifically to create one.
If your process requires an air atmosphere (oxygen), your physics are capped. You cannot use graphite or tungsten elements, as they will destroy themselves at high temperatures. You are effectively limited to MoSi₂ elements and a ceiling of roughly 1800°C.
If you need to go higher—into the realm of 3000°C for graphitization or advanced aerospace testing—you must abandon air. You must invest in complex systems that maintain a vacuum or pump inert gases like argon.
High temperature is not just about power. It is about environmental control.
The Psychology of the Redline
In finance, there is a concept called the "Margin of Safety." In engineering, we call it "Derating."
Every furnace has a Rated Continuous Operating Temperature. This is the redline.
Can you drive a car at the redline for 10 hours straight? Yes. Should you? Absolutely not.
Running a furnace at its absolute maximum rating drastically shortens the lifespan of the heating elements and the insulation. It stresses the ceramic fiber and pushes the quartz or alumina tubes to their breaking points.
The Rule of Thumb: Always buy a furnace rated for 50°C to 100°C higher than your actual process temperature.
If you need to sinter at 1150°C daily, do not buy a 1200°C furnace. Buy a 1400°C model. You pay for the capability, but you are buying reliability.
Selecting Your Tier
We can simplify the complexity of thermal engineering into three distinct tiers of application.
1. The Standard Tier (Up to 1200°C)
The Solution: FeCrAl (Kanthal) Elements. The Use Case: This covers the majority of lab needs—drying, binder burnout, and general synthesis. It is cost-effective and low-maintenance.
2. The High-Temperature Tier (1200°C to 1800°C)
The Solution: SiC or MoSi₂ Elements. The Use Case: Essential for advanced materials science. This is where you sinter metal powders, process advanced ceramics, and grow crystals.
3. The Ultra-High Tier (Above 1800°C)
The Solution: Graphite or Refractory Metals (in vacuum/inert gas). The Use Case: Specialized industrial research. Carbon fiber production and extreme material testing.
Summary of Capabilities
| Temperature Range | Heating Element | Typical Applications |
|---|---|---|
| Up to 1200°C | Kanthal (FeCrAl) | Drying, binder burnout, heat treatment |
| 1200°C - 1800°C | Silicon Carbide (SiC) / MoSi₂ | Advanced ceramics, sintering, crystal growth |
| Above 1800°C | Graphite (Inert/Vacuum only) | Graphitization, aerospace research |
Conclusion: Matching the Tool to the Task
A higher temperature rating is not always "better." It is simply different, usually more expensive, and often more complex to operate.
The goal is not to buy the furnace with the highest number on the datasheet. The goal is to buy the furnace that solves your specific problem with the highest degree of reliability.
At KINTEK, we don't just sell equipment; we sell solutions to thermal problems. Whether you need a robust daily driver for annealing or a specialized vacuum system for ultra-high temperature research, our engineers understand the trade-offs.
Stop guessing about the limits of your equipment. Let us help you calculate the Margin of Safety your research requires.
Visual Guide
Related Products
- Rotary Tube Furnace Split Multi Heating Zone Rotating Tube Furnace
- Laboratory Vacuum Tilt Rotary Tube Furnace Rotating Tube Furnace
- 1200℃ Split Tube Furnace with Quartz Tube Laboratory Tubular Furnace
- 1400℃ Laboratory High Temperature Tube Furnace with Alumina Tube
- 1700℃ Laboratory High Temperature Tube Furnace with Alumina Tube
Related Articles
- Rotary Furnaces: Advanced Materials Processing and Applications
- Unlocking Efficiency: The Ultimate Guide to Rotary Tube Furnaces
- Rotary Furnaces: A Comprehensive Guide to Advanced Materials Processing
- Exploring the Advantages of Rotary Tube Furnaces in Industrial and Laboratory Applications
- Exploring Rotary Tube Furnaces: A Comprehensive Guide




















