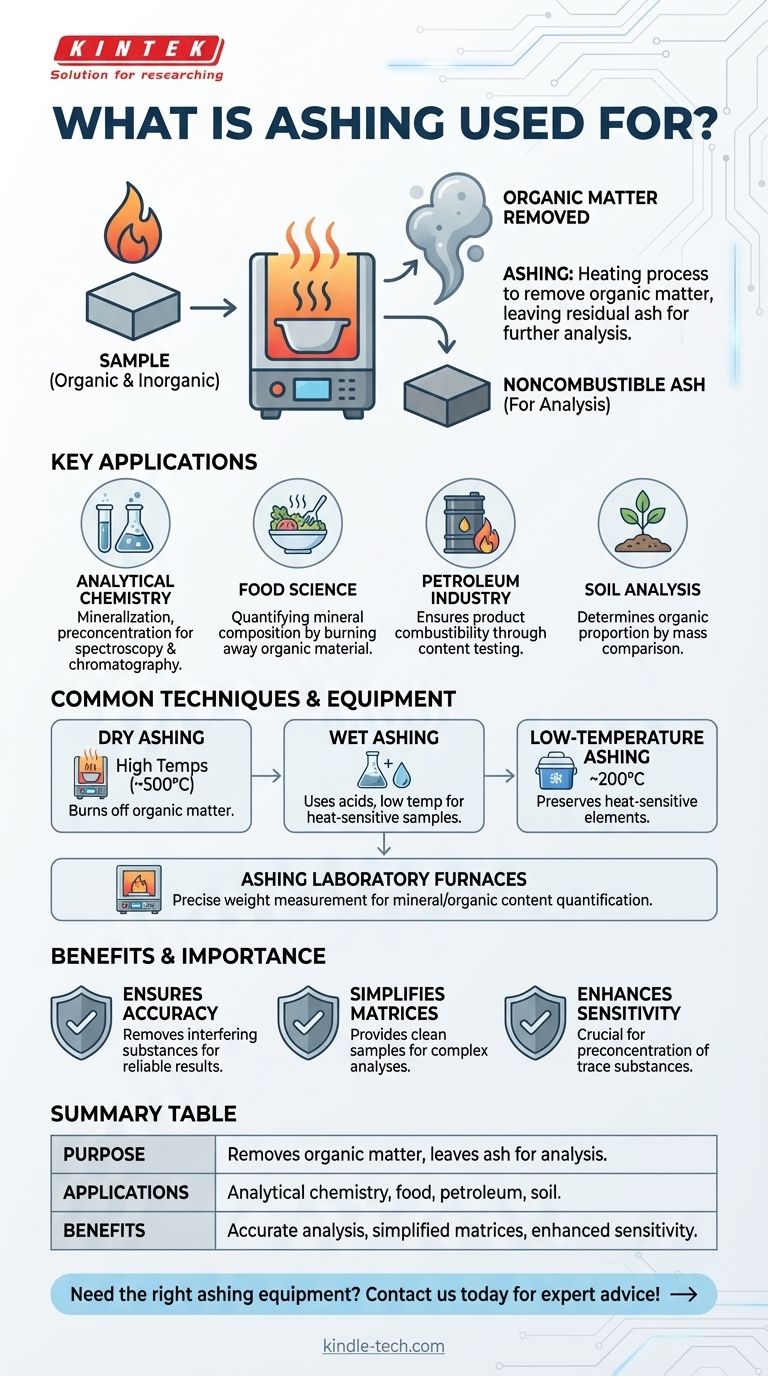
Ashing is a critical process used in various industries and scientific fields to remove organic matter and other interfering components from samples, leaving behind noncombustible ash for further analysis. It is commonly employed in analytical chemistry, food science, petroleum testing, and soil analysis. Techniques such as dry ashing, wet ashing, and low-temperature ashing are used depending on the sample type and analytical requirements. Ashing helps quantify mineral compositions, ensure product combustibility, and prepare samples for techniques like spectroscopy and chromatography. It is also essential in preconcentration of trace substances and determining organic proportions in materials like soil.

Key Points Explained:
-
Definition and Purpose of Ashing
- Ashing is a process that involves heating a sample to remove organic matter and other interfering matrix constituents, leaving behind noncombustible ash.
- It is primarily used to prepare samples for accurate analysis by eliminating components that could interfere with measurements.
-
Applications of Ashing
- Analytical Chemistry: Ashing is used for mineralization and preconcentration of trace substances before chemical or optical analysis, such as chromatography or spectroscopy.
- Food Science: It helps quantify the mineral composition of food samples by burning away organic materials.
- Petroleum Industry: Ashing ensures the combustibility of petroleum products by testing their contents.
- Soil Analysis: It determines the organic proportion of soil by comparing mass before and after ashing.
-
Common Ashing Techniques
- Dry Ashing: Involves heating samples at high temperatures (around 500°C) to burn off organic matter.
- Wet Ashing: Uses acids instead of high temperatures to remove organic components, suitable for heat-sensitive samples.
- Low-Temperature Ashing: Conducted at lower temperatures (around 200°C) to preserve heat-sensitive elements.
- Sulfated Ashing: Neutralizes and removes sulfur dioxide from samples.
- Closed System Ashing: Uses airtight chambers to control the atmosphere during the process, ensuring precision.
-
Role in Graphite Furnace Atomic Absorption (AA)
- Ashing is a crucial step in graphite furnace AA programs, where it removes matrix constituents that could interfere with the measurement of the analyte.
- This step ensures accurate and reliable results in elemental analysis.
-
Importance in Sample Preparation
- Ashing simplifies sample matrices, making it easier to analyze the residual ash for elemental composition.
- It is particularly useful in preconcentration of trace substances, enhancing the sensitivity of analytical techniques.
-
Equipment Used in Ashing
- Ashing Laboratory Furnaces: Designed to measure weight changes in samples as organic components are burnt away.
- These furnaces are essential for industries requiring precise quantification of mineral or organic content.
-
Benefits of Ashing
- Ensures accurate analysis by removing interfering substances.
- Provides a clean sample matrix for techniques like spectroscopy and chromatography.
- Helps in quantifying mineral content and organic proportions in various materials.
By understanding these key points, equipment and consumable purchasers can better appreciate the importance of ashing in sample preparation and analysis, ensuring they select the right tools and techniques for their specific needs.
Summary Table:
| Aspect | Details |
|---|---|
| Purpose | Removes organic matter, leaving noncombustible ash for analysis. |
| Applications | Analytical chemistry, food science, petroleum testing, soil analysis. |
| Techniques | Dry ashing, wet ashing, low-temperature ashing, sulfated ashing. |
| Equipment | Ashing laboratory furnaces for precise weight measurement. |
| Benefits | Ensures accurate analysis, simplifies sample matrices, enhances sensitivity. |
Need the right ashing equipment for your lab? Contact us today for expert advice and solutions!
Visual Guide
Related Products
- High Temperature Muffle Oven Furnace for Laboratory Debinding and Pre Sintering
- Graphite Vacuum Furnace High Thermal Conductivity Film Graphitization Furnace
- Ultra-High Temperature Graphite Vacuum Graphitization Furnace
- Vertical High Temperature Graphite Vacuum Graphitization Furnace
- Large Vertical Graphite Vacuum Graphitization Furnace
People Also Ask
- What role does a high-temperature muffle furnace play in the synthesis of manganese/cobalt-modified ceramic catalysts?
- What is the role of a muffle furnace in magnetite pellet roasting? Optimize Mineral Phase & Compressive Strength
- Why is the annealing treatment in a high-temperature muffle furnace critical for the preparation of an Sb-SnO2 interlayer?
- What conditions does a muffle furnace provide for molten salt energy storage? Expert Simulation for CSP Environments
- Why is a high-temp muffle furnace essential for ZnO-WO3 & ZnO-BiOI? Optimize Heterojunction Catalyst Performance



















