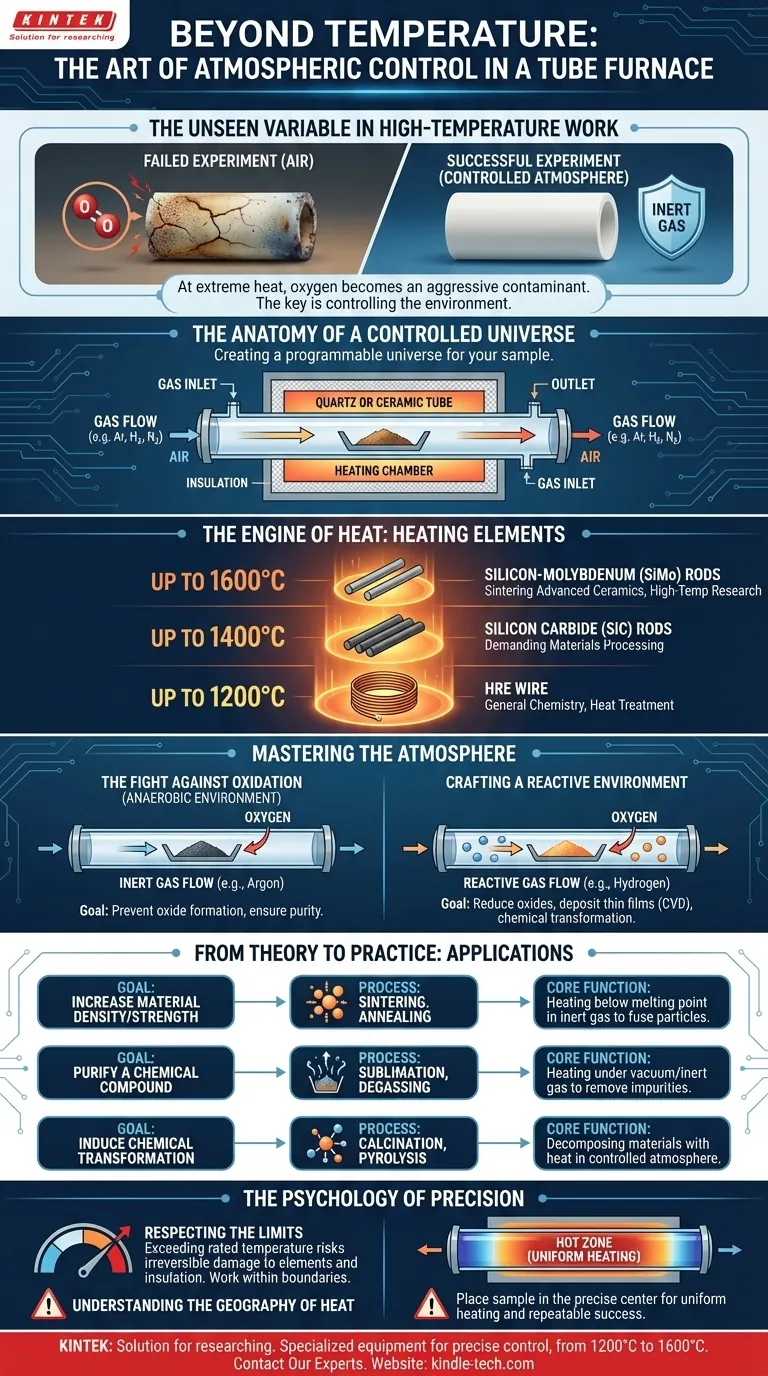
The Unseen Variable in High-Temperature Work
Imagine a materials scientist meticulously preparing a sample for sintering. The goal is to create a new ceramic alloy with exceptional hardness. The process requires heating the material to 1500°C, a temperature where molecules vibrate with incredible energy, ready to bond in new ways.
But after hours of careful heating, the sample emerges brittle and discolored. The experiment failed.
The culprit wasn't the temperature. It was the air. At such extreme heat, oxygen—normally life-giving—becomes a hyper-aggressive agent of contamination, ruining the sample's purity. This common laboratory tragedy reveals a fundamental truth: in advanced material processing, the most important question isn't "How hot can it get?" but rather, "How perfectly can I control the environment?"
The Anatomy of a Controlled Universe
A tube furnace appears simple: a tube running through a heating chamber. But this simplicity is deceptive. It is a highly engineered instrument designed to create a small, programmable universe for a sample.
The Engine of Heat
The maximum temperature of that universe is dictated by its engine: the heating element. The material choice here is a direct trade-off between cost, performance, and the laws of physics.
- Up to 1200°C: Furnaces with HRE (High-Resistance Element) wire are the reliable workhorses for general chemistry and heat treatment.
- Up to 1400°C: Reaching this next tier requires silicon carbon rods, opening the door to more demanding materials processing.
- Up to 1600°C: The frontier of many lab applications is reached with silicon-molybdenum rods, essential for sintering advanced ceramics and high-temperature research where new material properties are born.
Each material represents a different level of technological capability, a key that unlocks a new range of experimental possibilities.
Why Atmosphere Is Everything
The real genius of the tube furnace, however, lies in its ability to banish the air.
By sealing the tube and flowing a specific gas—like inert argon or reactive hydrogen—through it, an operator takes complete command of the chemical environment. This isn't just a feature; it is the core advantage that separates a tube furnace from a simple oven.
The Fight Against Oxidation
For many processes, the primary goal is to create an oxygen-free (anaerobic) environment. This prevents the formation of unwanted oxides, ensuring the chemical and structural purity of the final product. It is the difference between creating a flawless material and an expensive piece of contaminated waste.
Crafting a Reactive Environment
Conversely, some processes require a specific atmosphere to proceed. A stream of hydrogen might be used to reduce a metal oxide, or a specific gas mixture could be necessary to deposit a thin film on a substrate (Chemical Vapor Deposition). The furnace becomes an active reactor, not just a passive heater.
From Theory to Practice: The Furnace at Work
This dual mastery of heat and atmosphere makes the tube furnace indispensable for a wide range of critical tasks. Its applications are defined by the specific problem it solves.
| Goal | Process Enabled | Core Function |
|---|---|---|
| Increase Material Density/Strength | Sintering, Annealing | Heating materials below melting point in an inert gas to fuse particles together. |
| Purify a Chemical Compound | Sublimation, Degassing | Heating a substance under vacuum or inert gas to remove volatile impurities or gases. |
| Induce a Chemical Transformation | Calcination, Pyrolysis | Decomposing materials with heat in a controlled atmosphere to create new compounds. |
The Psychology of Precision
Operating such a powerful tool demands a certain mindset. The temptation to push a machine "just a little bit higher" than its rated temperature is a common human impulse, but in this context, it's a catastrophic one.
Respecting the Limits
Exceeding the maximum rated temperature doesn't just risk a failed experiment; it can cause irreversible damage to the heating elements and insulation. The limit isn't a suggestion; it is a physical boundary defined by the materials themselves. True mastery comes from working with precision within those boundaries.
Understanding the Geography of Heat
Similarly, the "rated temperature" applies to the center of the tube—the "hot zone." The temperature naturally falls off toward the ends. An experienced operator understands this thermal geography and places the sample in the precise center to guarantee uniform heating. It’s a subtle but critical detail that separates repeatable success from frustrating inconsistency.
Choosing the right furnace, then, is not about buying the one with the highest number on the dial. It's about deeply understanding your process and selecting a tool that provides the exact combination of temperature and atmospheric control you need to achieve your goal. It's a choice about precision, purity, and possibility.
At KINTEK, we provide the specialized equipment that enables this level of control, from versatile 1200°C furnaces for general applications to high-performance 1600°C systems for cutting-edge research. To ensure your work is built on a foundation of reliability and precision, Contact Our Experts.
Visual Guide
Related Products
- Rotary Tube Furnace Split Multi Heating Zone Rotating Tube Furnace
- Laboratory Vacuum Tilt Rotary Tube Furnace Rotating Tube Furnace
- 1200℃ Split Tube Furnace with Quartz Tube Laboratory Tubular Furnace
- 1400℃ Laboratory High Temperature Tube Furnace with Alumina Tube
- 1700℃ Laboratory High Temperature Tube Furnace with Alumina Tube
Related Articles
- Exploring the Functions and Advantages of Rotary Tube Furnaces
- Rotary Furnaces: A Comprehensive Guide to Advanced Materials Processing
- Exploring the Benefits and Applications of Rotary Furnaces: A Comprehensive Guide
- Maximize Rotary Furnace Performance: Design Advancements for Efficient Material Processing
- The Geometry of Heat: Why Motion Matters in Material Synthesis




















