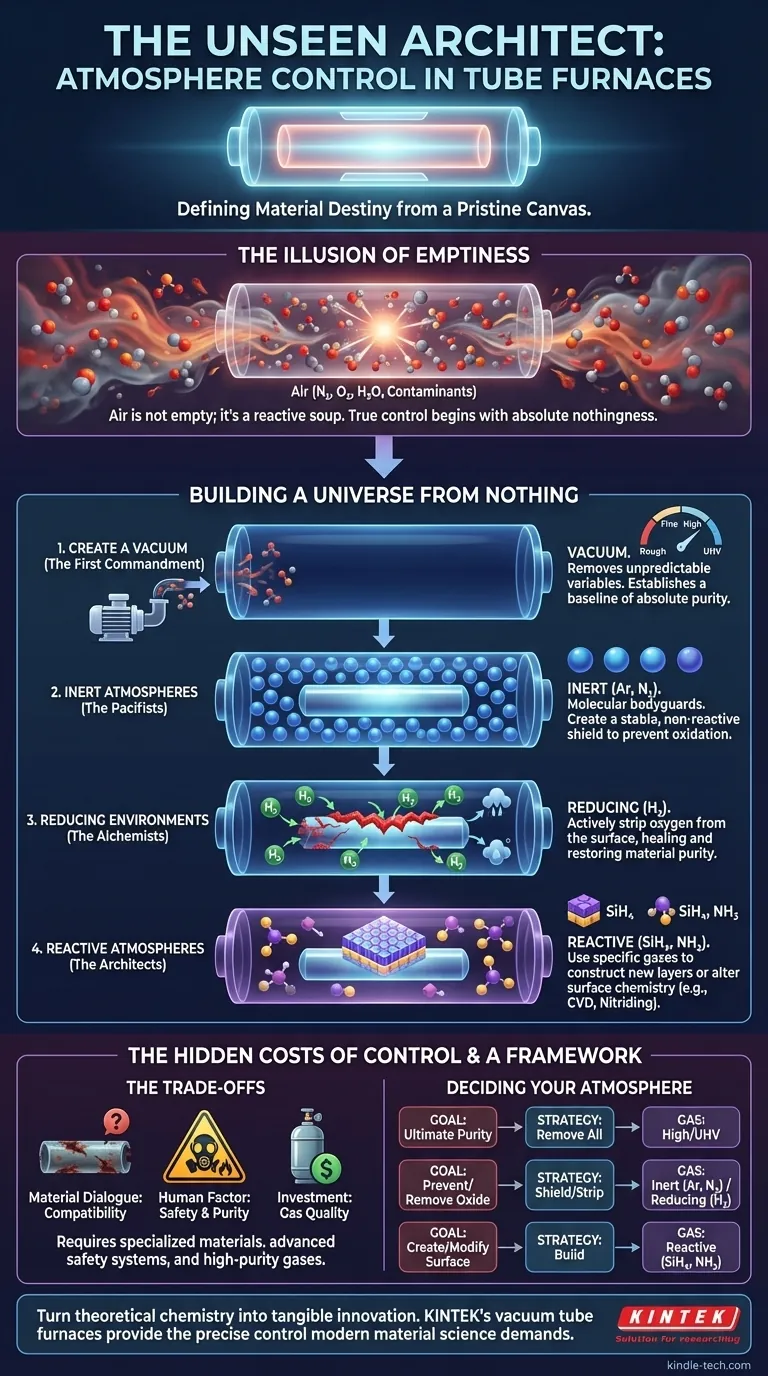
The Illusion of Emptiness
In material science, the most dangerous enemy is often invisible. The air we breathe, a seemingly benign mix of nitrogen and oxygen, becomes a violently reactive agent at high temperatures. For an engineer synthesizing a novel alloy or a researcher growing a perfect crystal, ambient air is not empty space; it's a chaotic soup of contaminants waiting to ruin a week's worth of work.
This is the first psychological hurdle to overcome: realizing that true control begins not with adding something, but with taking everything away. The goal is to create a pocket of absolute nothingness—a perfect vacuum—and from that pristine canvas, build a new, precisely controlled world for your material to be born in.
Building a Universe from Nothing
A vacuum tube furnace is more than a heater; it's a small-scale universe creator. Its power lies in its ability to first achieve a vacuum, setting the stage for whatever controlled atmosphere a process demands.
The First Commandment: Create a Vacuum
Evacuation is the non-negotiable first step. By removing the air, you eliminate the unpredictable variables—oxygen, moisture, and other trace gases. This isn't just cleaning; it's establishing a baseline of absolute purity. The quality of this "nothingness" can be tailored:
- Rough & Fine Vacuum: Sufficient for many processes where minimal oxidation is the goal.
- High & Ultra-High Vacuum (UHV): Essential for sensitive applications like semiconductor research or thin-film deposition, where even a single stray molecule can be a critical defect.
The Pacifists: Inert Atmospheres
Once the void is created, the first impulse is often to protect it. Inert gases are molecular bodyguards. Backfilling the tube with Argon or Nitrogen creates a stable, non-reactive shield around the sample.
These gases don't participate in the reaction; their job is to simply be present, preventing the chaos of the outside world from creeping back in and stopping unwanted reactions like oxidation. Helium, with its high thermal conductivity, can even help by speeding up heating and cooling cycles.
The Alchemists: Reducing Environments
Sometimes, prevention isn't enough. A material might already have a thin layer of oxide on its surface. This is where reducing atmospheres come in. They are the alchemists of material processing.
Introducing a controlled mixture of Hydrogen (H₂) actively strips oxygen atoms from the material's surface, healing it and restoring it to a pure, metallic state. It’s a process of purification that actively reverses damage, ensuring the final material is chemically pristine.
The Architects: Reactive Atmospheres
The final level of control is not just to protect or purify, but to build. Reactive atmospheres use specific gases as building blocks to construct new layers or alter the material's surface chemistry.
In processes like Chemical Vapor Deposition (CVD), gases like Silane (SiH₄) are introduced to decompose at high temperatures, depositing a thin film of silicon. In nitriding, Ammonia (NH₃) is used to harden the surface of a metal. This is material architecture at the atomic scale, and it demands equipment that can handle these often-corrosive building blocks, such as furnaces equipped with quartz or alumina tubes.
The Hidden Costs of Control
Mastering atmospheric control is a game of trade-offs, where every choice has consequences.
The Material Dialogue
The furnace components must be compatible with the chosen atmosphere. A reactive gas like ammonia will attack a standard metallic tube, leading to equipment failure and sample contamination. This requires a deep understanding of material compatibility—a dialogue between your process gas and your furnace. Choosing a system with the correct tube and sealing technology is paramount.
The Human Factor
The power of gases like Hydrogen and Carbon Monoxide comes with responsibility. Their flammability and toxicity demand respect, requiring advanced safety interlocks, ventilation, and gas detection systems. Similarly, the purity of your gas source is a direct investment in the quality of your result. Skimping on gas purity is a bet against your own success.
A Framework for Deciding Your Atmosphere
Your process goal dictates your atmospheric choice. The logic is straightforward:
| Desired Outcome | Atmospheric Strategy | Common Gases |
|---|---|---|
| Ultimate Purity | Remove all reactive molecules. | High/Ultra-High Vacuum |
| Oxidation Prevention | Shield the sample with a non-reactive buffer. | Argon (Ar), Nitrogen (N₂) |
| Oxide Removal/Reduction | Actively strip oxygen from the surface. | Hydrogen (H₂) |
| Surface Creation/Modification | Introduce specific atoms to build or alter the surface. | Silane (SiH₄), Ammonia (NH₃) |
Achieving this level of control requires not just knowledge, but the right instrumentation. KINTEK's vacuum tube furnaces are designed to provide the precise, reliable atmospheric control that modern material science demands, from UHV to complex reactive gas mixtures. They are the tools that turn theoretical chemistry into tangible innovation. To find the right furnace for the universe you need to build, Contact Our Experts
Visual Guide
Related Products
- Vacuum Sealed Continuous Working Rotary Tube Furnace Rotating Tube Furnace
- Laboratory High Pressure Vacuum Tube Furnace
- Laboratory Vacuum Tilt Rotary Tube Furnace Rotating Tube Furnace
- Split Chamber CVD Tube Furnace with Vacuum Station Chemical Vapor Deposition System Equipment Machine
- 1200℃ Controlled Atmosphere Furnace Nitrogen Inert Atmosphere Furnace
Related Articles
- The Architecture of Emptiness: Why the Void is the Ultimate Material Shield
- Exploring the Functions and Advantages of Rotary Tube Furnaces
- Exploring Rotary Tube Furnaces: A Comprehensive Guide
- The Pursuit of Purity: How a Sealed Vacuum Furnace Creates Metallurgical Certainty
- Unlocking Efficiency: The Ultimate Guide to Rotary Tube Furnaces




















