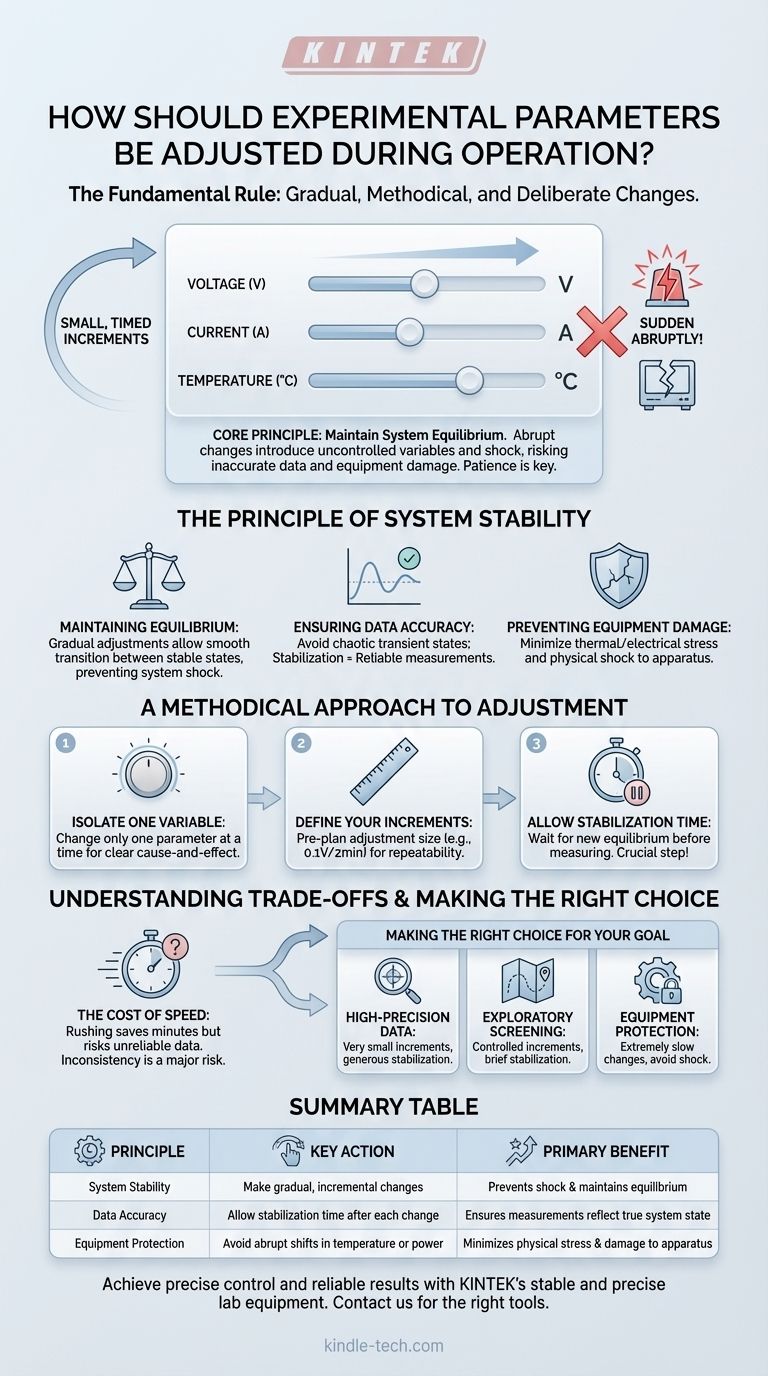
The fundamental rule for adjusting experimental parameters during operation is to do so gradually and methodically. Whether modifying voltage, current, or temperature, changes must be made in small, deliberate increments, never abruptly. This approach is essential for protecting the integrity of your results and the longevity of your equipment.
The core principle is to maintain system equilibrium. Abrupt changes introduce uncontrolled variables and thermal or electrical shock, leading to inaccurate data and potential equipment damage. A patient, gradual approach is the bedrock of reliable and repeatable experimental work.

The Principle of System Stability
Any experiment in operation exists in a state of relative equilibrium. Your goal is to move the system from one stable state to another in a controlled way. Rapid, large-scale changes disrupt this control and compromise your work.
Maintaining Equilibrium
A running experiment, such as an electrochemical cell in a constant temperature bath, is a balanced system. Each parameter is interrelated. A sudden spike in voltage or a rapid drop in temperature doesn't just change one variable; it sends a shock through the entire system.
The system needs time to respond and settle into a new, stable state. Gradual adjustments allow for this transition to happen smoothly, ensuring that the conditions you measure are the actual, stable conditions of the experiment.
Ensuring Data Accuracy
Measurements taken immediately after a sudden parameter change are often meaningless. They reflect a chaotic, transient state, not a true data point.
By adjusting in small steps and allowing the system to stabilize after each one, you can be confident that every measurement you record accurately reflects the system's behavior under that specific set of conditions.
Preventing Equipment Damage
Sudden changes can cause significant physical stress to your apparatus. A rapid increase in current can generate intense localized heat, potentially damaging sensitive electrodes or components inside a cell.
Similarly, an abrupt temperature shift can cause materials to crack or warp due to thermal shock. Gradual adjustments minimize these physical stresses, protecting your investment and preventing costly downtime.
A Methodical Approach to Adjustment
Adopting a structured process for changing parameters is non-negotiable for serious experimental work. It moves you from "turning knobs" to conducting controlled, scientific inquiry.
Isolate One Variable at a Time
Whenever possible, your experimental design should only alter one parameter at a time. If you change both voltage and temperature simultaneously, you cannot determine which variable was responsible for the observed change in your results.
Define Your Increments
Before you begin, decide on the size of your adjustments. For example, you might plan to "increase voltage by 0.1V every two minutes" or "raise the bath temperature by 2°C every ten minutes." This makes your process repeatable.
Allow for Stabilization Time
This is the most critical step. After making an incremental adjustment, you must wait for the system to reach a new equilibrium before taking a measurement. The required time will vary by system, but it is an essential part of the process that cannot be skipped.
Understanding the Trade-offs
While the gradual approach is superior, it's important to understand the practical implications and why people are often tempted to rush.
The Cost of Speed
The primary trade-off is time. A methodical, gradual process is inherently slower than making large, fast changes. However, rushing an experiment to save a few minutes often invalidates the hours of work by producing unreliable data. The time saved is an illusion if the results are unusable.
The Risk of Inconsistency
Manual, abrupt changes are difficult to reproduce with any consistency. A "quick twist of the dial" is not a quantifiable action. A process built on small, timed increments is repeatable by you or any other operator, which is the foundation of sound scientific and engineering work.
Making the Right Choice for Your Goal
Your specific approach can be tailored to the objective of your experiment, but the core principle of gradual change remains constant.
- If your primary focus is high-precision data for analysis: Use very small increments and allow for generous stabilization times to ensure maximum accuracy and minimal system noise.
- If your primary focus is exploratory screening of conditions: You may use slightly larger, yet still controlled, increments to cover a wider range more quickly, while still allowing for a brief stabilization period.
- If your primary focus is protecting delicate or expensive equipment: Prioritize extremely slow and gradual changes, especially for temperature and power, to completely avoid any risk of thermal or electrical shock.
Ultimately, disciplined control over your experimental parameters is what separates random observation from rigorous, meaningful investigation.
Summary Table:
| Principle | Key Action | Primary Benefit |
|---|---|---|
| System Stability | Make gradual, incremental changes | Prevents shock and maintains equilibrium |
| Data Accuracy | Allow for stabilization time after each change | Ensures measurements reflect true system state |
| Equipment Protection | Avoid abrupt shifts in temperature or power | Minimizes physical stress and damage to apparatus |
Achieve precise control and reliable results in your lab. Proper parameter adjustment is fundamental to successful experimentation. KINTEK specializes in providing high-quality lab equipment and consumables designed for stability and precision. Whether you need a reliable furnace for controlled heating or precise instrumentation for electrochemical work, our products are built to support your methodical approach. Contact us today to find the right tools to enhance your experimental integrity and protect your investment. Get in touch with our experts
Visual Guide