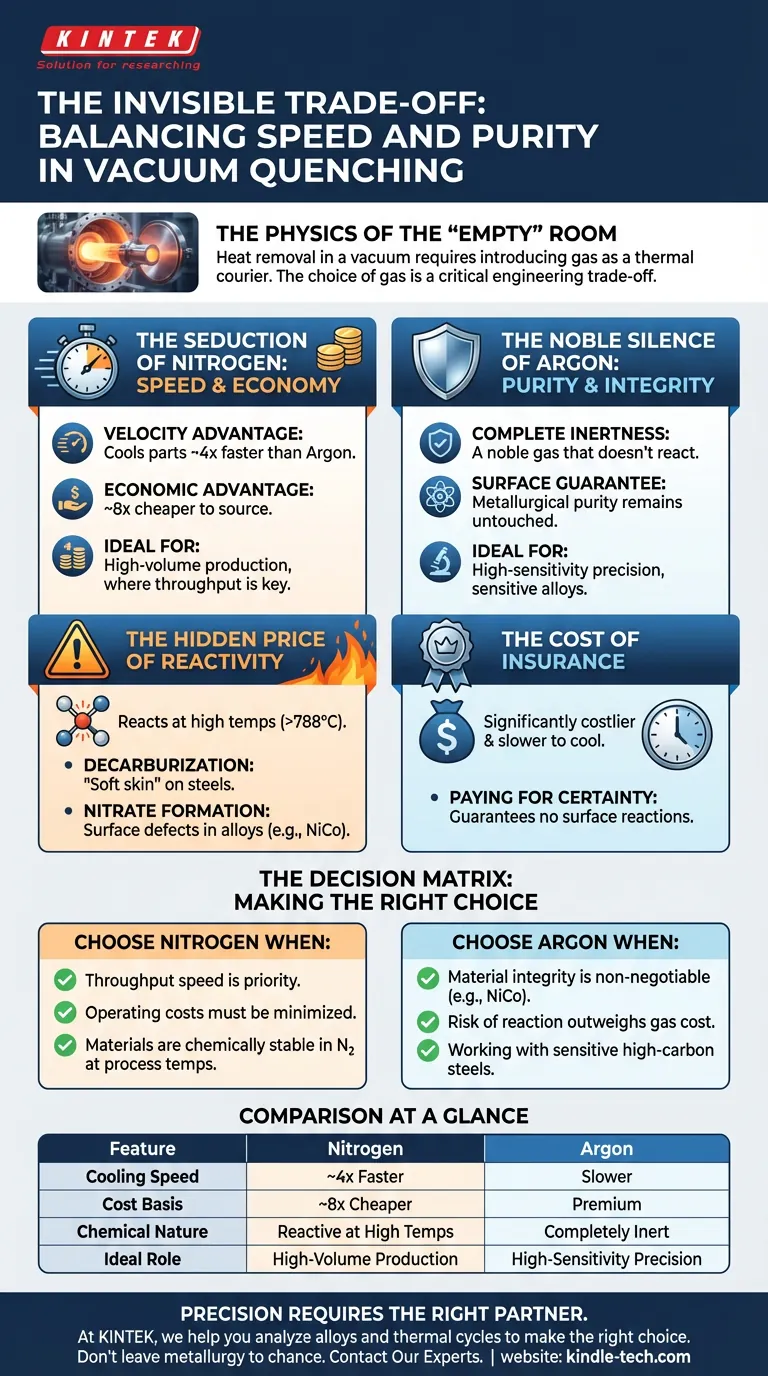
The Physics of the "Empty" Room
In the precise world of vacuum furnaces, heat is both the tool and the enemy.
Creating heat is straightforward. Removing it—rapidly and uniformly—is where the engineering art begins. In a vacuum, you lose the natural benefit of convection. Heat is stranded, escaping only through slow radiation.
To force a phase change in metallurgy, we often need violence. We need to "quench" the material, shocking it from high temperatures back to stability to lock in specific mechanical properties.
To do this, we break the vacuum. We introduce gas to act as a thermal courier, carrying heat away from the part and into the heat exchanger.
The question isn't whether to use gas. The question is: Which gas?
The industry typically offers two contenders: Nitrogen and Argon. The choice between them is a classic study in risk management.
The Seduction of Nitrogen: Speed and Economy
If you look purely at the spreadsheet, Nitrogen is the undisputed champion. It appeals to our desire for efficiency and throughput.
Nitrogen is a heavy lifter. Due to its physical properties, it facilitates convection far better than its noble counterpart.
- The Velocity Advantage: Nitrogen cools parts approximately four times faster than Argon.
- The Economic Advantage: It is roughly eight times cheaper to source.
For high-volume production, these numbers are hard to ignore. When cycle time dictates profit margins, Nitrogen is the default choice. It is aggressive, efficient, and abundant.
But efficiency often hides a cost.
The Hidden Price of Reactivity
The problem with Nitrogen is that it is not truly "inert."
It plays the part of a neutral gas well at room temperature. But push the environment to 1450°F (788°C) or higher, and Nitrogen begins to wake up. It stops being a coolant and starts becoming a chemical reactant.
This is the engineering equivalent of a side effect in medicine. You cure the thermal problem, but you introduce a chemical pathology.
The Risks
- Decarburization in Steels: Nitrogen can react with surface carbon, stripping it away. This leaves a "soft skin" on the part, altering the hardness you fought so hard to achieve.
- Nitrate Formation in Alloys: For Nickel-Cobalt (NiCo) alloys, high-temperature Nitrogen is dangerous. It reacts to form nitrates on the surface.
In aerospace or medical applications, where failure is not an option, a surface reaction is a structural defect.
The Noble Silence of Argon
Argon is the stoic of the periodic table.
It is a noble gas, meaning it possesses complete chemical inertness. It does not care how hot your furnace gets. It does not care what alloy you are processing.
It enters the chamber, moves the heat, and leaves without leaving a trace.
This silence is expensive. Argon is slower to cool and significantly costlier to buy. However, you aren't paying for speed; you are paying for insurance.
By using Argon, you guarantee that the metallurgical purity of the part’s surface remains exactly as the chemist intended, untouched by the atmosphere used to cool it.
The Decision Matrix
Engineering is rarely about perfect solutions; it is about the right set of trade-offs.
When selecting your quenching gas, you must weigh the "Cost of Goods Sold" against the "Cost of Failure."
Choose Nitrogen when:
- Throughput speed is the priority.
- Operating costs must be minimized.
- You are processing materials that are chemically stable in nitrogen at your specific process temperatures.
Choose Argon when:
- Material integrity is non-negotiable (e.g., NiCo alloys).
- The risk of surface reaction outweighs the cost of the gas.
- You are working with high-carbon steels sensitive to decarburization.
Comparison at a Glance
| Feature | Nitrogen | Argon |
|---|---|---|
| Cooling Speed | ~4x Faster | Slower |
| Cost Basis | ~8x Cheaper | Premium |
| Chemical Nature | Reactive at High Temps | Completely Inert |
| Ideal Role | High-Volume Production | High-Sensitivity Precision |
Precision Requires the Right Partner
A vacuum furnace is only as good as the decisions made by its operator. Using the wrong gas can lead to scrapped parts, costly rework, and compromised structural integrity.
At KINTEK, we understand that lab equipment and consumables are not just commodities—they are vital components of your scientific success. We specialize in navigating these technical trade-offs.
Whether you need to maximize throughput or ensure absolute surface purity, our team can help you analyze your specific alloys and thermal cycles to make the right choice.
Don't leave your metallurgy to chance. Contact Our Experts
Visual Guide
Related Products
- Vacuum Cold Mounting Machine for Sample Preparation
- Vacuum Induction Melting Spinning System Arc Melting Furnace
- Vacuum Heat Treat Sintering Brazing Furnace
- Vacuum Heat Treat and Pressure Sintering Furnace for High Temperature Applications
- Vacuum Heat Treat Furnace and Levitation Induction Melting Furnace
Related Articles
- Defeating the Void: The Quiet Power of Vacuum Hot Pressing
- Comprehensive Overview of Vacuum Evaporation Systems
- Lessons Learned from a Laboratory Vacuum Pump Oil Change Fire Incident
- Techniques for Sample Preparation in X-Ray Fluorescence Analysis: Pressed and Loose Powder Methods
- The Architecture of Emptiness: Why Vacuum Level is a Decision, Not Just a Number




















