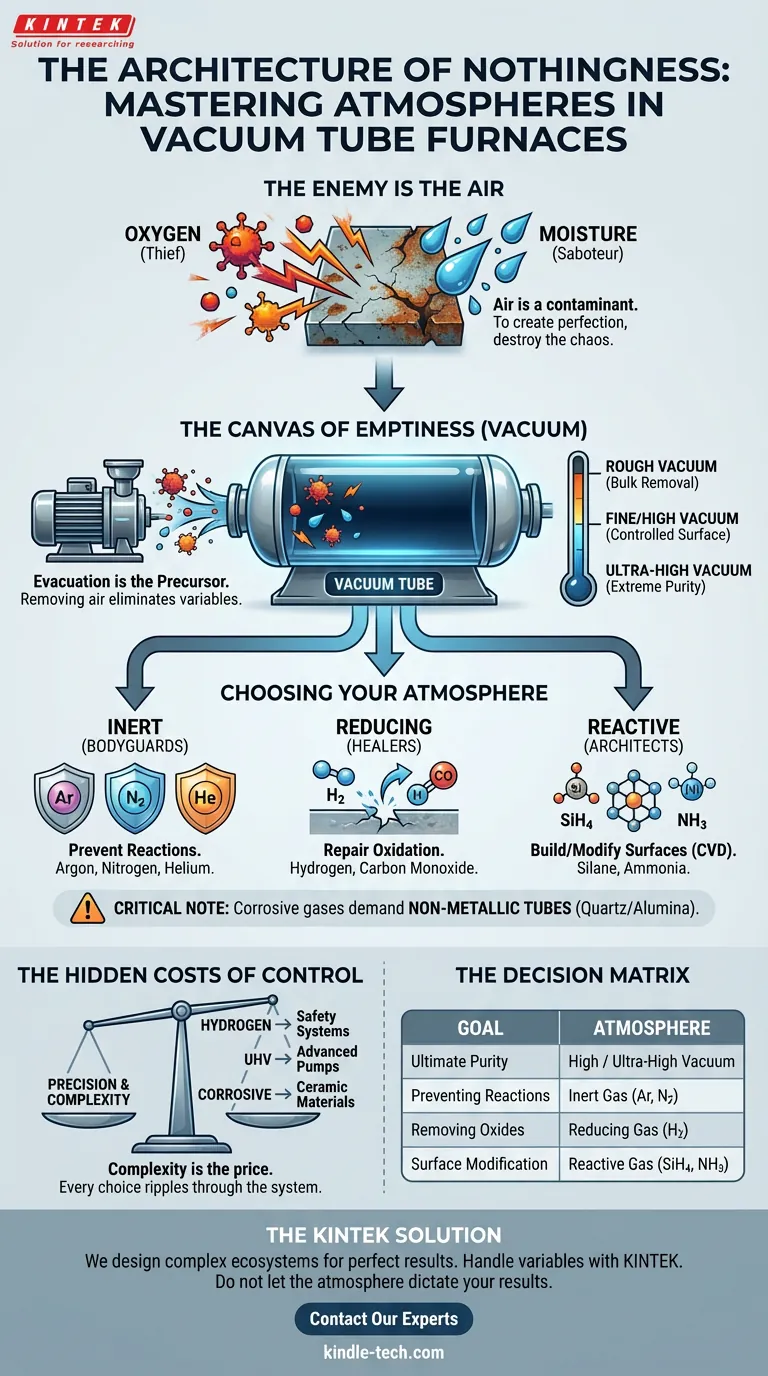
The Enemy is the Air
In the delicate world of materials science, the ambient atmosphere is not a resource. It is a contaminant.
Oxygen is a thief. It steals electrons and degrades surfaces. Moisture is a saboteur. It introduces microscopic defects that ruin structural integrity.
To create something perfect—whether it is a semiconductor, a new alloy, or a ceramic composite—you first need to destroy the chaos of the room you are standing in.
This is the primary function of the vacuum tube furnace. It is not merely an oven; it is a machine designed to impose order on entropy.
The Canvas of Emptiness
Before you can paint, you must have a clean canvas. In thermal processing, this step is evacuation.
The defining capability of these furnaces is their ability to strip away the atmosphere entirely. This is the "Precursor." By removing air, we eliminate the variables that cause failure.
But "empty" is a relative term. The level of emptiness you achieve dictates the purity of your result:
- Rough Vacuum: Removes the bulk of air, sufficient for basic non-sensitive work.
- Fine to High Vacuum: Required when surface reactions must be strictly controlled.
- Ultra-High Vacuum: The domain of extreme purity, where even a few stray molecules are too many.
Once the chamber is evacuated, the furnace becomes a vessel of potential. You can leave it empty, or you can fill it with intention.
Choosing Your Atmosphere
Once the vacuum has scrubbed the stage clean, you can introduce specific actors to play out the chemical reaction.
The Bodyguards: Inert Environments
When you want heat without chemistry, you use inert gases. They are the chaperones of the atomic world. They ensure that atoms vibrate and settle without bonding to anything they shouldn't.
- Argon: The heavy lifter. Cost-effective and thermally conductive.
- Nitrogen: The standard choice, provided your material doesn’t react with it.
- Helium: The sprinter. Its high thermal conductivity allows for rapid heating and cooling cycles.
The Healers: Reducing Environments
Sometimes, the material is already damaged (oxidized). You need an atmosphere that actively repairs the surface.
- Hydrogen (H₂): The aggressive healer. It strips oxygen from surfaces. However, it demands respect—and rigorous safety protocols.
- Carbon Monoxide (CO): Effective, but toxic. It requires careful handling.
The Architects: Reactive Environments
In advanced processes like Chemical Vapor Deposition (CVD), the gas isn't there to protect or heal. It is there to build.
- Silane (SiH₄): Essential for depositing silicon films.
- Ammonia (NH₃): Used for nitriding, hardening the surface of the material.
A Critical Note on Hardware: You cannot simply pump corrosive gases into a standard furnace. Reactive environments demand non-metallic tubes (like quartz or alumina). If you use standard metal components here, the gas will eat the machine before it processes your sample.
The Hidden Costs of Control
Complexity is the price of precision.
Operating a vacuum tube furnace is a study in trade-offs. Every choice you make ripples through the rest of the system.
If you choose Hydrogen, you are not just buying gas. You are investing in safety interlocks, explosion-proof fittings, and ventilation systems. The cost of safety is non-negotiable.
If you choose Ultra-High Vacuum, you are changing your pumping infrastructure. A simple roughing pump won't cut it; you need turbo-molecular or cryogenic systems.
If you choose Corrosive Gases, you are limiting your structural materials. You trade the durability of metal for the chemical resistance of ceramics.
The Decision Matrix
Great engineering is about matching the tool to the problem.
We see many researchers struggle not because their chemistry is wrong, but because their environmental control is mismatched. Here is how to simplify the decision:
| If your goal is... | Then your atmosphere is... |
|---|---|
| Ultimate Purity | High or Ultra-High Vacuum |
| Preventing Reactions | Inert Gas (Argon, Nitrogen) |
| Removing Oxides | Reducing Gas (Hydrogen) |
| Surface Modification (CVD) | Reactive Gas (Silane, Ammonia) |
The KINTEK Solution
A vacuum tube furnace is more than a heated tube. It is a complex ecosystem of pumps, seals, controllers, and safety valves.
When this ecosystem works, it fades into the background, leaving you with nothing but perfect results. When it fails, it becomes the only thing you can think about.
At KINTEK, we design our equipment with the understanding that "nothingness" is hard to achieve. Whether you need the absolute silence of a high vacuum or the precise chemistry of a reactive mixture, our systems are built to handle the variables so you don't have to.
Do not let the atmosphere dictate your results.
Visual Guide
Related Products
- Vacuum Sealed Continuous Working Rotary Tube Furnace Rotating Tube Furnace
- Laboratory High Pressure Vacuum Tube Furnace
- Laboratory Vacuum Tilt Rotary Tube Furnace Rotating Tube Furnace
- Split Chamber CVD Tube Furnace with Vacuum Station Chemical Vapor Deposition System Equipment Machine
- 1200℃ Controlled Atmosphere Furnace Nitrogen Inert Atmosphere Furnace
Related Articles
- The Geometry of Heat: Why Motion Matters in Material Synthesis
- Unlocking Efficiency: Exploring the Advantages and Applications of Rotary Tube Furnaces
- The Pursuit of Purity: How a Sealed Vacuum Furnace Creates Metallurgical Certainty
- Exploring the Advantages of Rotary Tube Furnaces in Industrial and Laboratory Applications
- Exploring Rotary Tube Furnaces: A Comprehensive Guide




















