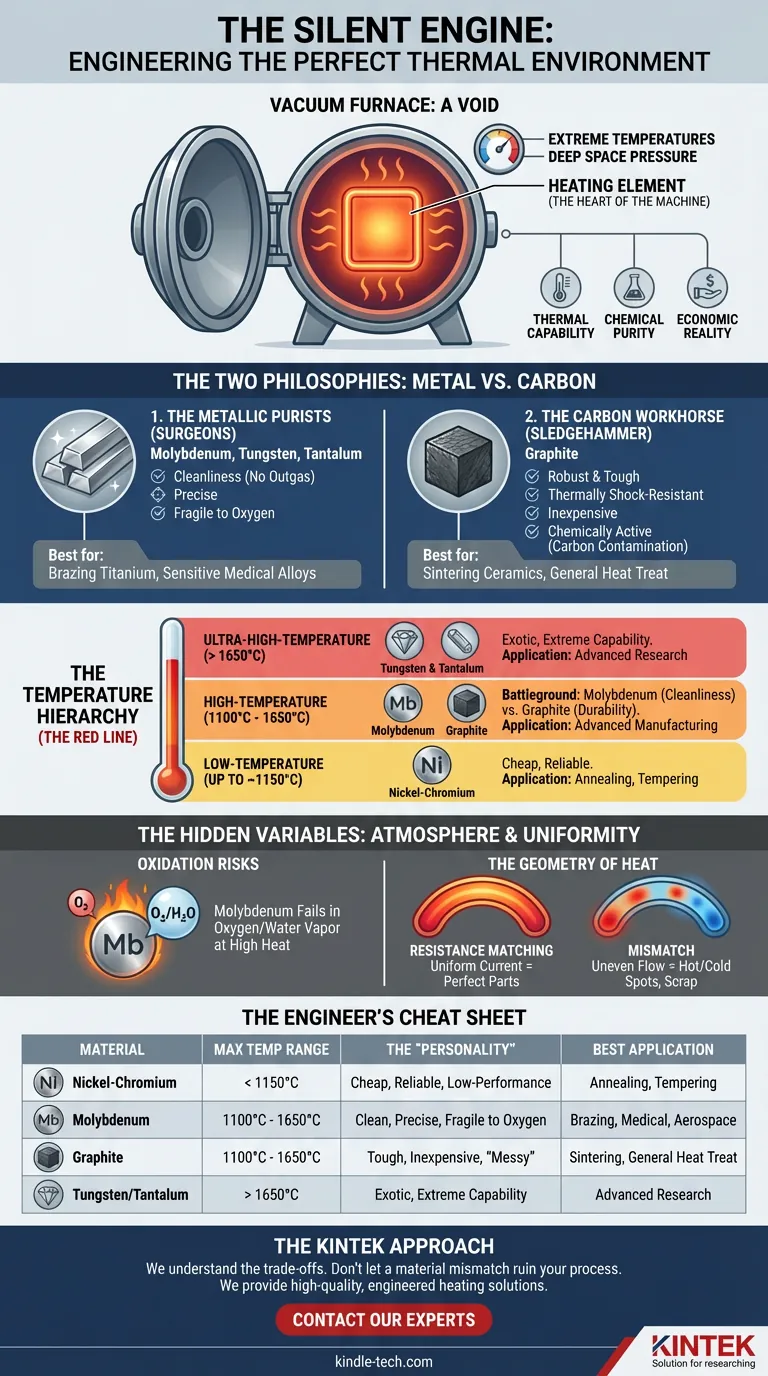
The Architecture of Heat
A vacuum furnace is a contradiction. It is a vessel designed to contain nothing—a void—yet it is responsible for forging the strongest materials on earth.
Inside this void, the environment is violent. Temperatures soar to levels where ordinary matter disintegrates. The pressure drops to mimic deep space.
At the center of this chaos sits the heating element.
It is the heart of the machine. If the vacuum pump is the lungs, the heating element is the pulse. It dictates what is possible. It determines whether a medical implant remains sterile or an aerospace component fails under stress.
Choosing this component is not a shopping task. It is a critical engineering decision that balances three opposing forces: thermal capability, chemical purity, and economic reality.
The Two Philosophies: Metal vs. Carbon
When engineers design a "hot zone," they generally choose between two distinct material philosophies. Each has its own personality, strengths, and fatal flaws.
1. The Metallic Purists (Molybdenum, Tungsten, Tantalum)
These are the surgeons of the thermal world.
Metallic elements are prized for their cleanliness. They do not outgas. They do not shed particles. If you are brazing titanium or processing sensitive medical alloys, metal is often the only choice.
- Molybdenum: The industry standard for high-purity work (1100°C to 1650°C). It is stable and clean.
- Tungsten & Tantalum: The heavy lifters. When temperatures exceed 1650°C—approaching the melting point of common ceramics—these refractory metals are required. They are expensive, brittle, and brilliant.
- Nickel-Chromium: The accessible option. Useful only for lower temperatures (up to ~1150°C), typically for annealing or tempering.
2. The Carbon Workhorse (Graphite)
Graphite is the sledgehammer. It is robust, thermally shock-resistant, and significantly cheaper than refractory metals.
However, graphite is chemically active. In high temperatures, it creates a carbon-rich environment. For sintering ceramics, this is often fine. For processing certain steel alloys, it is a disaster. The carbon will migrate into the metal, altering its metallurgy and ruining the batch.
The Temperature Hierarchy
In engineering, temperature is not just a number on a dial. It is a threshold of material failure.
The selection of a heating element is dictated primarily by the "red line"—the point of no return.
Low-Temperature (Up to ~1150°C)
Here, the stakes are lower. Nickel-Chromium alloys dominate. They are cost-effective and reliable. But like a budget car engine, if you redline them consistently, they will degrade rapidly.
High-Temperature (1100°C to 1650°C)
This is the standard operating range for most advanced manufacturing. The battle here is between Molybdenum and Graphite.
- Choose Molybdenum for cleanliness (aerospace, medical).
- Choose Graphite for durability and cost savings (sintering, general heat treat).
Ultra-High-Temperature (Above 1650°C)
This is rarefied air. Only Tungsten and Tantalum survive here. These materials are difficult to fabricate and expensive to procure, but for research and advanced material science, they are irreplaceable.
The Hidden Variables: Atmosphere and Uniformity
A heating element does not exist in a vacuum—metaphorically speaking. It interacts with everything around it.
Oxidation Risks: A Molybdenum element is a marvel of engineering, but it has an Achilles' heel. A trace amount of oxygen or water vapor at high heat will cause it to oxidize and fail catastrophically. Graphite, conversely, is immune to this specific failure mode but carries the risk of carbon contamination.
The Geometry of Heat: It is not enough to generate heat; you must control it.
For large hot zones, particularly those using graphite, resistance matching is vital. If the curved elements do not have identical electrical resistance, the current will flow unevenly. This creates hot spots and cold spots.
The result? A batch of parts where half are perfect and half are scrap.
Summary: The Engineer's Cheat Sheet
| Material | Max Temp Range | The "Personality" | Best Application |
|---|---|---|---|
| Nickel-Chromium | < 1150°C | Cheap, reliable, low-performance | Annealing, Tempering |
| Molybdenum | 1100°C - 1650°C | Clean, precise, fragile to oxygen | Brazing, Medical, Aerospace |
| Graphite | 1100°C - 1650°C | Tough, inexpensive, "messy" | Sintering, General Heat Treat |
| Tungsten/Tantalum | > 1650°C | Exotic, extreme capability | Advanced Research |
The KINTEK Approach
There is a difference between buying a spare part and investing in process reliability.
At KINTEK, we understand that a heating element is not just a wire or a rod. It is the defining variable of your laboratory's success. Whether you need the surgical purity of Molybdenum or the rugged durability of Graphite, the choice involves trade-offs that affect your budget and your data.
We help you navigate those trade-offs. We provide high-quality, engineered heating solutions designed to withstand the violent environment of the vacuum chamber, ensuring your science remains sound.
Don't let a material mismatch ruin your process. Contact Our Experts
Visual Guide
Related Products
- Silicon Carbide SiC Thermal Heating Elements for Electric Furnace
- Vacuum Heat Treat Furnace with Ceramic Fiber Liner
- Molybdenum Vacuum Heat Treat Furnace
- Molybdenum Disilicide (MoSi2) Thermal Elements Electric Furnace Heating Element
- Vacuum Heat Treat and Molybdenum Wire Sintering Furnace for Vacuum Sintering
Related Articles
- Understanding Electric Heating Systems(1): How Electric Furnaces Work and Their Advantages
- Precautions for installing silicon carbide stick
- Exploring the Benefits of Using Tungsten for Furnace Heating
- Selecting Heating Elements for Vacuum Furnaces
- The Invisible Variable: Selecting the Heart of Your Vacuum Furnace




















