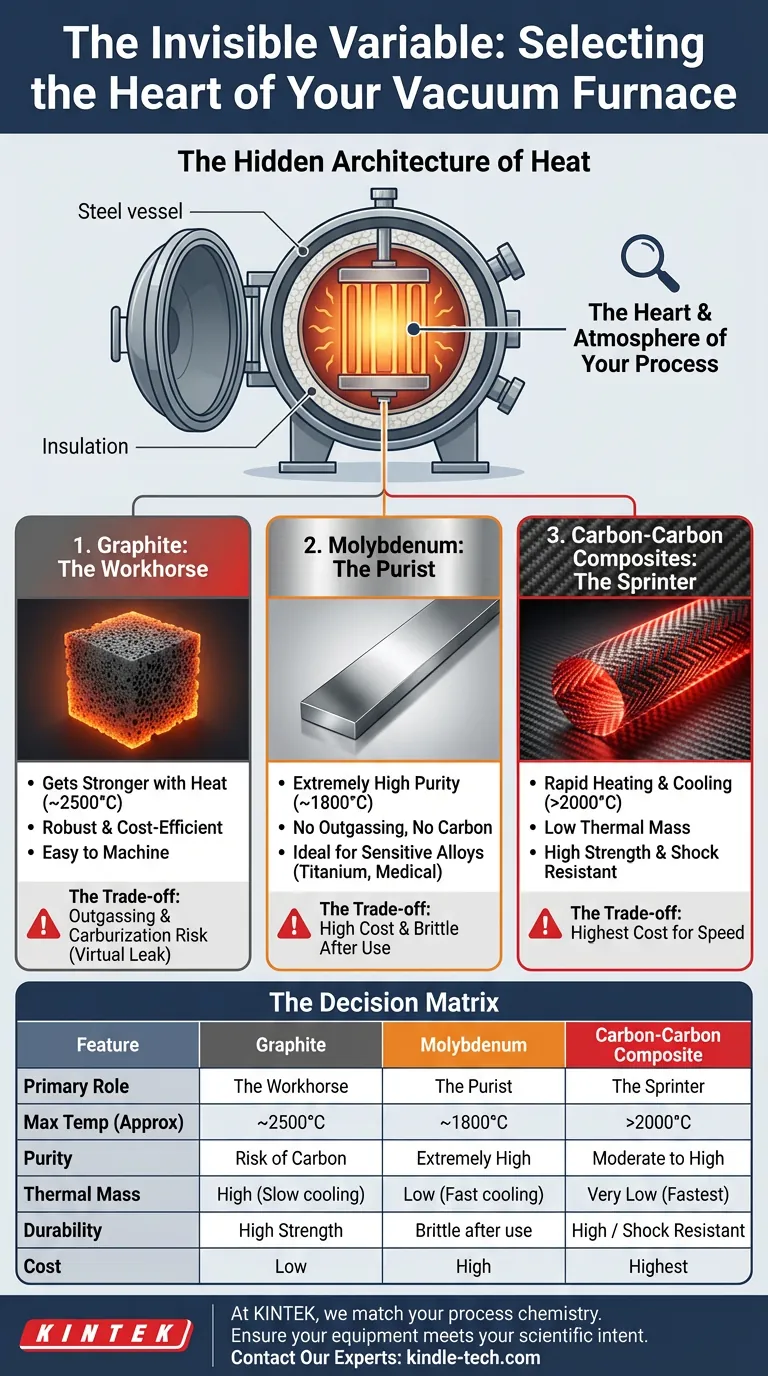
The Hidden Architecture of Heat
In engineering, what you cannot see is often more critical than what you can.
A vacuum furnace appears to be a static steel vessel from the outside. But inside, during a cycle, it is a dynamic, violent environment where temperature and chemistry collide.
At the center of this storm is the heating element.
It is easy to think of the heating element as a simple utility—a glorified lightbulb filament that just needs to get hot. This is a mistake. The material you choose for your heating element is not just a component; it is the atmosphere of your process.
It dictates the purity you can achieve. It defines the speed at which you can work. It determines whether your medical alloy is life-saving or scrap metal.
When designing or selecting a vacuum furnace, you are essentially choosing between three distinct philosophies: the rugged utility of Graphite, the clinical purity of Molybdenum, or the high-speed performance of Carbon-Carbon Composites.
Here is how to navigate the trade-offs.
The Physics of the Void
Vacuum furnaces operate in an oxygen-free environment. This is the only reason we can use the materials we do. In open air, graphite would burn away, and molybdenum would oxidize instantly.
The vacuum protects the element. But the element must also protect the workload.
The interaction between these two defines your success.
1. Graphite: The Paradox of Strength
Graphite is the standard for a reason. It is the workhorse of the heat-treating industry.
It possesses a rare, almost romantic engineering quality: it gets stronger as it gets hotter. Up to about 2500°C (4532°F), graphite defies the usual rules of material degradation. It is robust, relatively inexpensive, and easy to machine into complex geometries.
But graphite breathes.
Because it is porous, graphite acts like a sponge. When the furnace is open to air, the element absorbs moisture and gases. When the cycle begins and the heat rises, the element releases these trapped molecules—a process known as outgassing.
The Trade-off: You get incredible durability and cost-efficiency. In exchange, you must manage the "virtual leak" of outgassing, which can extend your pump-down times.
Furthermore, graphite is carbon. If you are processing materials that are chemically hungry for carbon (like Titanium), a graphite element can cause "carburization," subtly altering the surface chemistry of your parts.
2. Molybdenum: The Clinical Approach
If graphite is the sledgehammer, Molybdenum (Moly) is the scalpel.
In industries like aerospace and medical device manufacturing, contamination is not an inconvenience; it is a failure. You cannot have stray carbon atoms migrating into a hip implant or a jet engine turbine blade.
For these applications, we turn to refractory metals.
Molybdenum provides an all-metal, exceptionally clean hot zone. It does not outgas like graphite. It does not shed carbon. It offers the purest possible vacuum environment.
The Trade-off: Purity comes at a price—literally and physically. Molybdenum is significantly more expensive than graphite.
It is also temperamental. After repeated cycles of extreme heat, Molybdenum recrystallizes and becomes brittle. It loses its ductility. A graphite rod can take a knock; a used Molybdenum strip can shatter like glass if mishandled during maintenance. It demands respect and a steady hand.
3. Carbon-Carbon Composites: Buying Time
Time is the most expensive variable in any lab or production floor.
Traditional graphite is heavy. It has high thermal mass, meaning it soaks up a tremendous amount of heat. Once you turn the power off, a heavy graphite hot zone acts like a heat battery, continuing to radiate warmth long after you want the furnace to cool down.
Enter Carbon-Carbon Composites (CFC).
By reinforcing a graphite matrix with carbon fibers, engineers created a material that is stronger than standard graphite but significantly thinner and lighter.
The Trade-off: CFC elements hold very little heat. They allow the furnace to heat up rapidly and, more importantly, cool down immediately. This slashes cycle times, increasing throughput.
The downside is the upfront investment. You are paying a premium for speed and mechanical resilience.
The Decision Matrix
There is no "best" material. There is only the material that aligns with your specific constraints.
Great engineering is about understanding the failure mode you are willing to accept to get the performance you need.
Summary of Characteristics
| Feature | Graphite | Molybdenum | Carbon-Carbon Composite |
|---|---|---|---|
| Primary Role | The Workhorse | The Purist | The Sprinter |
| Max Temp (Approx) | ~2500°C | ~1800°C | >2000°C |
| Purity | Risk of Carbon dust/vapor | Extremely High | Moderate to High |
| Thermal Mass | High (Slow cooling) | Low (Fast cooling) | Very Low (Fastest cooling) |
| Durability | High Strength | Brittle after use | High / Shock Resistant |
| Cost | Low | High | Highest |
How to Choose
When you speak to a KINTEK expert, we start with the application, not the catalogue.
- Select Graphite if your process is general-purpose (hardening steels, brazing) and cost is a primary driver. It is reliable and forgiving.
- Select Molybdenum if your workload is chemically sensitive. If you are processing Titanium, Niobium, or Tantalum, you cannot risk carbon interaction. The element must be metal.
- Select Composites if your bottleneck is time. If you need to run multiple cycles per shift, the rapid cooling of CFC pays for itself in throughput.
The Human Element in Lab Equipment
A vacuum furnace is a system. If one variable is misaligned—if the element fights the workload—the system fails.
At KINTEK, we specialize in identifying these variables before they become problems. We understand that you aren't just buying a heating element; you are buying a controlled environment for your research or production.
Whether you need the ruggedness of graphite or the precision of molybdenum, our role is to ensure your equipment matches your scientific intent.
Do not leave your process chemistry to chance.
Visual Guide
Related Products
- Silicon Carbide SiC Thermal Heating Elements for Electric Furnace
- Molybdenum Disilicide (MoSi2) Thermal Elements Electric Furnace Heating Element
- Vacuum Heat Treat Furnace with Ceramic Fiber Liner
- 2200 ℃ Graphite Vacuum Heat Treat Furnace
- Vacuum Hot Press Furnace Heated Vacuum Press Machine Tube Furnace
Related Articles
- The Silent Engine: Engineering the Perfect Thermal Environment
- Precautions for installing silicon carbide stick
- Selecting Heating Elements for Vacuum Furnaces
- Why Your High-Temperature Furnace Elements Fail: The Critical Difference in Silicon Carbide
- Understanding Electric Heating Systems(1): How Electric Furnaces Work and Their Advantages




















