While not typically catalysts themselves, carbon nanotubes (CNTs) are used in catalyst systems primarily as high-performance supports for catalytic nanoparticles. Their unique physical and electronic properties dramatically enhance the efficiency, stability, and longevity of the actual catalyst material that is anchored to their surface.
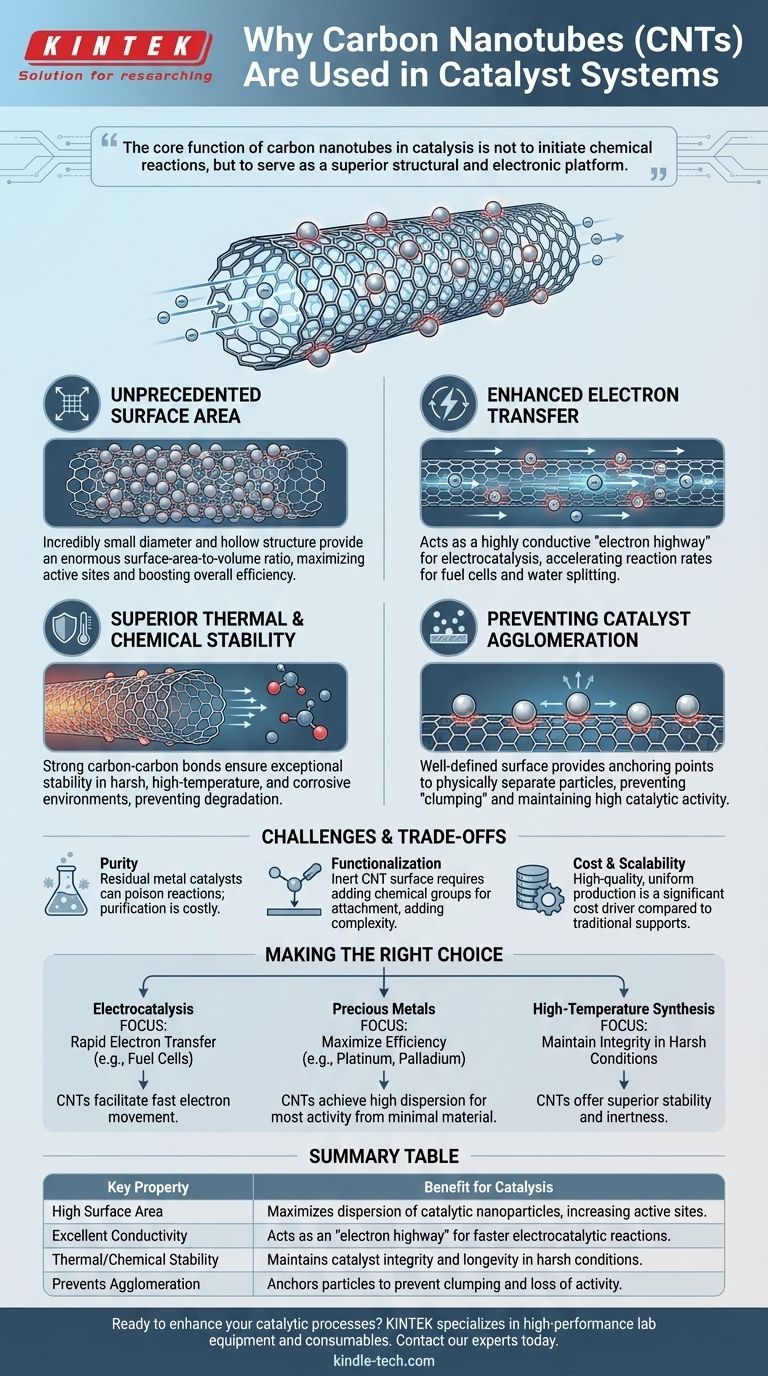
The core function of carbon nanotubes in catalysis is not to initiate chemical reactions, but to serve as a superior structural and electronic platform. They amplify the performance of catalytic particles by providing massive surface area, excellent conductivity, and robust stability.

Why CNTs Excel as Catalyst Supports
The effectiveness of a catalyst often depends as much on its support structure as on the catalyst material itself. A support's job is to hold the active catalytic particles, keeping them separated and accessible to reactants. Carbon nanotubes perform this role exceptionally well for several key reasons.
Unprecedented Surface Area
A fundamental principle of catalysis is that more active sites lead to faster reactions. The incredibly small diameter and hollow structure of CNTs provide an enormous surface-area-to-volume ratio.
This allows for a very high and uniform dispersion of catalytic nanoparticles, maximizing the number of active sites available for chemical reactions and significantly boosting overall efficiency.
Enhanced Electron Transfer
For a large class of reactions known as electrocatalysis (critical for fuel cells and water splitting), the speed of electron transfer is paramount. CNTs are excellent electrical conductors.
When used as a support, they create a highly conductive network, acting as an "electron highway" that efficiently shuttles electrons to and from the active catalyst particles, accelerating the reaction rate.
Superior Thermal and Chemical Stability
Many industrial catalytic processes occur under harsh conditions, including high temperatures and corrosive chemical environments.
The strong carbon-carbon bonds in CNTs give them exceptional thermal stability and chemical inertness. This robust nature prevents the support material from degrading, ensuring the catalyst remains stable and active for a longer operational lifetime.
Preventing Catalyst Agglomeration
A common failure mode for catalysts is the aggregation or "clumping" of the active nanoparticles. When particles clump together, their effective surface area decreases, and catalytic activity plummets.
The well-defined surface of CNTs provides distinct anchoring points that physically separate the catalyst particles, preventing them from migrating and agglomerating even at high temperatures.
Understanding the Trade-offs and Challenges
While powerful, the use of CNTs in catalysis is not without its difficulties. A clear understanding of these limitations is essential for practical application.
The Challenge of Purity
The synthesis of CNTs often requires metal catalysts (like iron or nickel), which can remain as impurities in the final product. These residual metals can interfere with or poison the desired catalytic reaction, requiring extensive and costly purification steps.
Difficulties in Functionalization
The surface of a pristine carbon nanotube is relatively inert. To securely anchor catalyst nanoparticles, the CNT surface must often be "functionalized"—a process of adding chemical groups that act as attachment points. This adds complexity and cost to the manufacturing process.
Cost and Scalability
Producing high-quality, uniform carbon nanotubes at an industrial scale remains a significant cost driver. For many applications, the performance benefits of using CNT supports must be weighed against their higher cost compared to traditional materials like activated carbon or alumina.
Making the Right Choice for Your Goal
The decision to use CNTs as a catalyst support should be driven by the specific demands of the application.
- If your primary focus is electrocatalysis (e.g., fuel cells, water splitting): CNTs are invaluable for their ability to facilitate rapid electron transfer between the electrode and the catalyst.
- If your primary focus is maximizing efficiency with precious metals: Use CNTs to achieve a high dispersion of nanoparticles, ensuring you get the most activity from a minimal amount of expensive material like platinum or palladium.
- If your primary focus is high-temperature chemical synthesis: The exceptional thermal and chemical stability of CNTs makes them a superior support for maintaining catalyst integrity under harsh conditions.
Ultimately, leveraging carbon nanotubes as a support structure transforms a good catalyst into a high-performance, durable catalytic system.
Summary Table:
| Key Property | Benefit for Catalysis |
|---|---|
| High Surface Area | Maximizes dispersion of catalytic nanoparticles, increasing active sites. |
| Excellent Conductivity | Acts as an "electron highway" for faster electrocatalytic reactions. |
| Thermal/Chemical Stability | Maintains catalyst integrity and longevity in harsh conditions. |
| Prevents Agglomeration | Anchors particles to prevent clumping and loss of activity. |
Ready to enhance your catalytic processes? KINTEK specializes in high-performance lab equipment and consumables, including advanced catalyst supports. Our expertise can help you leverage materials like carbon nanotubes to achieve greater efficiency, stability, and longevity in your reactions. Contact our experts today to discuss how we can support your laboratory's specific catalyst development needs.
Visual Guide
Related Products
- Carbon Graphite Boat -Laboratory Tube Furnace with Cover
- Boron Nitride (BN) Ceramic Tube
- Hexagonal Boron Nitride HBN Thermocouple Protection Tube
People Also Ask
- What is the disadvantage of graphite furnace? Managing Reactivity and Contamination Risks
- Why is graphite used in furnaces? For Extreme Heat, Purity, and Efficiency
- Why graphite is used as a crucible to melt metal? Unlock Superior High-Temperature Performance
- What is the temperature range of graphite crucible? Choose the Right Crucible for Your High-Temp Application
- What metals can you melt in a graphite crucible? A Guide to Safe & Efficient Melting


