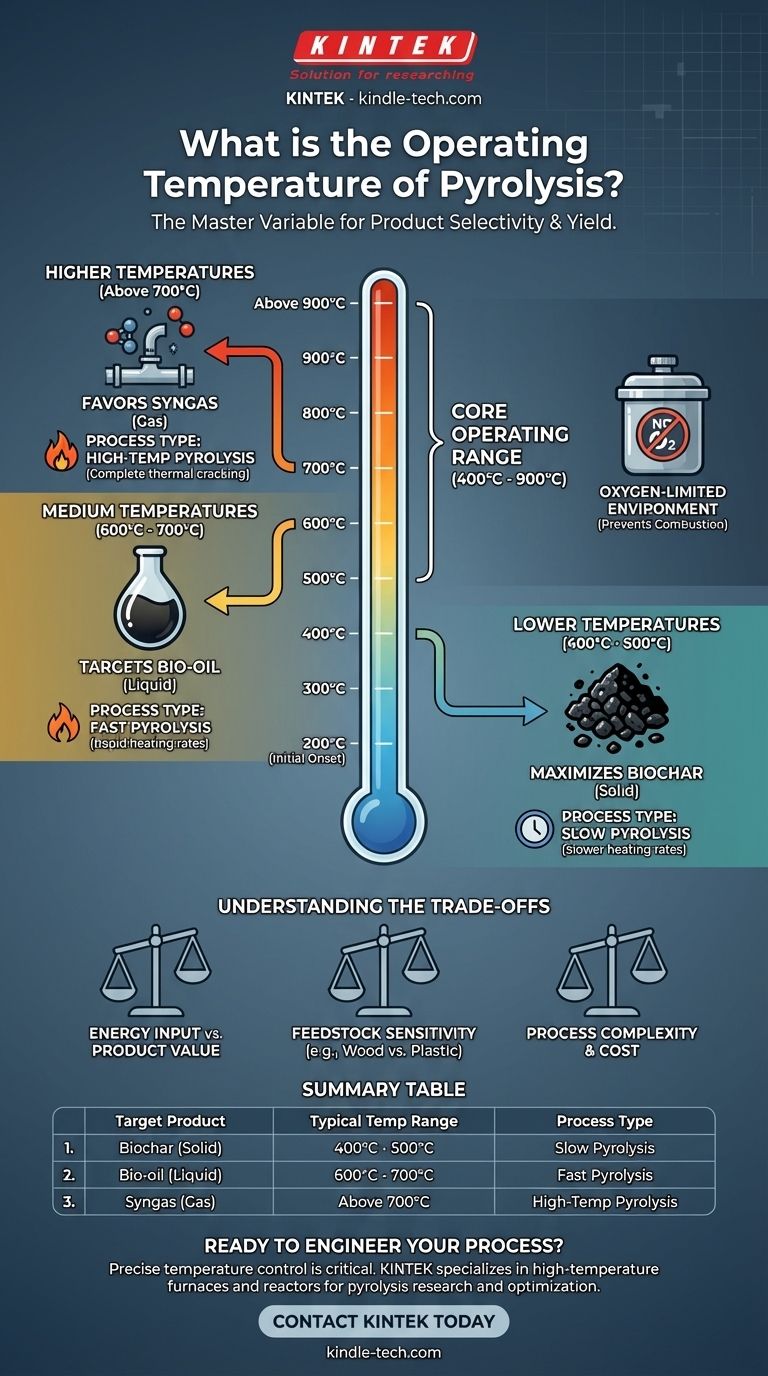
In short, the operating temperature for pyrolysis typically ranges from 400°C to 900°C (750°F to 1650°F). However, the process of thermal decomposition can begin at temperatures as low as 200°C, with the specific temperature being carefully selected based on the feedstock material and the desired end-products.
The core principle to understand is that pyrolysis temperature is not a single, fixed value. It is the primary control variable used to determine whether the final output will be a solid (biochar), a liquid (bio-oil), or a gas (syngas).

Why Temperature is the Master Variable
Pyrolysis is, by definition, the thermal decomposition of materials at elevated temperatures in an oxygen-limited environment. Temperature directly governs the speed and extent of this decomposition, making it the most critical parameter in any pyrolysis operation.
The Initial Onset of Pyrolysis
For many organic materials, like wood, the process begins at relatively low temperatures. The breakdown of weaker chemical bonds starts to occur between 200°C and 300°C. This is the initial stage where volatile compounds begin to release.
The Core Operating Range
Most industrial pyrolysis processes operate within a much higher and more effective range of 400°C to 900°C. Within this window, the feedstock is broken down efficiently into its valuable components.
The Critical Role of an Oxygen-Free Environment
It is crucial to understand that these temperatures are applied in an oxygen-limited or completely oxygen-free atmosphere. The absence of oxygen prevents combustion (burning) and instead forces the material to thermally break apart into different molecules.
How Temperature Determines the Final Product
The "why" behind selecting a specific temperature is to control the final products. Different temperatures favor different outputs, a concept known as product selectivity.
Lower Temperatures for Biochar (Solid)
Slow pyrolysis, which is often conducted at the lower end of the spectrum (around 400°C to 500°C), maximizes the production of biochar. The slower heating rates and lower temperatures leave more of the carbon behind in a stable, solid form.
Medium Temperatures for Bio-oil (Liquid)
Fast pyrolysis targets the production of bio-oil. By rapidly heating the material to a medium temperature range (around 600°C to 700°C), the process cracks the feedstock into vapors. These vapors can then be rapidly cooled and condensed into a liquid fuel.
Higher Temperatures for Syngas (Gas)
At the highest temperatures (typically above 700°C), the process favors the production of syngas (synthesis gas). These extreme conditions crack all the material, including the heavier vapors and biochar, into very simple, non-condensable gas molecules like hydrogen and carbon monoxide.
Understanding the Trade-offs
Choosing a pyrolysis temperature is an engineering decision that involves balancing competing factors. There is no single "best" temperature, only the optimal one for a specific goal.
Energy Input vs. Product Value
Achieving and maintaining higher temperatures requires a significantly greater energy input. This operational cost must be justified by the market value of the end-product, whether it's high-value syngas or specialty biochar.
Feedstock Sensitivity
Different feedstocks (e.g., wood, agricultural waste, plastics) decompose differently. A temperature that is optimal for producing bio-oil from wood may not be suitable for plastic, highlighting the need to tune the process for each specific material.
Process Complexity and Cost
Operating at very high temperatures in an oxygen-free environment demands sophisticated reactor technology and control systems. This increases the capital and maintenance costs of the facility.
Making the Right Choice for Your Goal
Your target temperature should be dictated entirely by the product you intend to create.
- If your primary focus is maximizing biochar production: Operate at the lower end of the temperature spectrum, typically 400°C to 500°C, using a slow pyrolysis process.
- If your primary focus is producing bio-oil (liquid fuel): Target a medium temperature range around 600°C with rapid heating rates, which is characteristic of fast pyrolysis.
- If your primary focus is generating syngas (gaseous fuel): Utilize the highest temperatures, generally above 700°C, to ensure complete thermal cracking of the feedstock into simple gases.
Ultimately, selecting the correct pyrolysis temperature is a strategic decision that directly engineers the desired chemical outcome.
Summary Table:
| Target Product | Typical Temperature Range | Process Type |
|---|---|---|
| Biochar (Solid) | 400°C - 500°C | Slow Pyrolysis |
| Bio-oil (Liquid) | 600°C - 700°C | Fast Pyrolysis |
| Syngas (Gas) | Above 700°C | High-Temp Pyrolysis |
Ready to Engineer Your Pyrolysis Process?
Selecting the precise temperature is critical for achieving your target product yield and quality. The right lab equipment is essential for research, development, and process optimization.
KINTEK specializes in high-temperature laboratory furnaces and reactors designed for precise pyrolysis applications. Our solutions provide the accurate temperature control and oxygen-free environments you need to reliably produce biochar, bio-oil, or syngas from your specific feedstock.
Let us help you build a more efficient and profitable process. Our experts are ready to discuss your requirements and recommend the ideal equipment for your laboratory.
Contact KINTEK today for a consultation and take control of your thermal conversion outcomes.
Visual Guide
Related Products
- Electric Rotary Kiln Small Rotary Furnace Biomass Pyrolysis Plant
- Electric Rotary Kiln Continuous Working Small Rotary Furnace Heating Pyrolysis Plant
- Electric Rotary Kiln Pyrolysis Furnace Plant Machine Calciner Small Rotary Kiln Rotating Furnace
- Customizable Laboratory High Temperature High Pressure Reactors for Diverse Scientific Applications
- High Pressure Laboratory Autoclave Reactor for Hydrothermal Synthesis
People Also Ask
- What are the principles of a rotary kiln? Master the Mechanics of High-Temperature Processing
- What is a rotary kiln reactor? A Guide to Industrial Thermal Processing
- What are the different types of calciners? A Guide to Selecting the Right Thermal Processing Equipment
- What is the temperature of a rotating kiln? It Depends on Your Material and Process Goal
- Is rotary kiln a furnace? Discover the Key Differences for Industrial Processing










